Detecting Peripheral Arterial Disease in Primary Care: A Population Based Study
Article information
Abstract
Background
Peripheral arterial disease (PAD) can progress silently without any clinical symptoms. Ankle-brachial index (ABI) is the recommended method used in primary care. We aimed to determine the prevalence of PAD and its related risk factors in primary care.
Methods
In this cross-sectional study, 250 participants aged ≥45 years were recruited randomly from the registered patients of family health center in a district of Izmir, Turkey. Patients’ demographic data, PAD symptoms, and PAD risk factors were obtained. The PAD group consisted of patients with ABI levels <0.9; the non-compressible artery (NCA) group consisted of patients with ABI levels >1.3.
Results
The prevalence of PAD was 17.6% (22.5% in women and 11.1% in men), while that of NCA was 15.2% (12.7% in women and 25.0% in men). About 27.3% and 54.5% of patients with PAD did not have claudication and problems with walking distance, respectively. Of the NCA patients, 15.8% had problems with walking distance and 39.5% had claudication. Regression analysis revealed two predictors of PAD (age ≥65 years: odds ratio [OR], 3.51; 95% confidence interval [CI], 1.65–7.47; claudication: OR, 3.41; 95% CI, 1.58–7.39) and three predictors of NCA (age <65 years: OR, 2.55; 95% CI, 1.01–6.45; male sex: OR, 2.40; 95% CI, 1.10–5.25; triglyceride [TG] >200 mg/dL: OR, 4.11; 95% CI, 1.58–10.67).
Conclusion
PAD had a prevalence of 17.6% and was associated with age ≥65 years and claudication. NCA had a prevalence of 15.2% and was associated with age <65 years, TG >200 mg/dL, and male sex.
INTRODUCTION
Peripheral arterial disease (PAD) is a progressive disease resulting in narrowing and blockage of the vessels [1]. PAD is a common condition with an associated morbidity and mortality. If it is left untreated, it can increase the risk of heart attack, stroke, amputation, and death [2]. Moreover, the clinical symptoms of PAD such as intermittent claudication, arterial ulcers, necrosis, and gangrene can greatly reduce patients’ quality of life [3]. Several diseases and conditions, including advanced age, smoking, diabetes, hypertension, hyperlipidemia, male sex, and elevated blood glucose, are considered as risk factors of PAD [2]. A low ankle-brachial index (ABI) level is an important indicator of atherosclerosis, but only 20% of the patients who have ABI level <0.90 develop symptoms.3) Therefore, PAD is currently considered as a subclinical disease, which can progress silently. Due to this fact, PAD must be diagnosed in primary health care centers [3].
In the United States, the prevalence of PAD is 2.5% in patients aged between 40 and 59 years, 8.3% in those aged between 60 and 69 years, and 18.8% in those aged between 70–79 years [4]. A prevalence study conducted in the Netherlands showed that the prevalence of PAD increases with age, and 19.1% of the PAD patients were aged over 55 years [5]. PAD prevalence was reported to be 19.76% and 20.0% in two different studies conducted in Turkey [6,7]. Although the number of studies in Turkey focusing on PAD prevalence are increasing in the recent years, prevalence studies especially population-based ones remain scarce.
This population-based study aimed to identify patients with PAD using the ABI method and their risk factors in order to determine high-risk patients.
METHODS
A cross-sectional survey was conducted among the patients at Balcova district of İzmir, which is the third largest city in Turkey. Using systematic random sampling method, 250 participants were chosen among 1,010 registered patients aged over 45 years from a family health center (FHC). The sample size was calculated with a 95% confidence interval and statistical power of 80%.
Data were collected via face-to-face interviews. A questionnaire, prepared by the researchers, included questions related to patients’ demographic data (sex and age), risk factors like comorbid diseases (such as diabetes mellitus [DM], hypertension, cerebrovascular disease [CVD], renal disease, chronic obstructive pulmonary disease [COPD], coronary artery disease [CAD], and hyperlipidemia), smoking habit, family history of heart disease and symptoms of PAD (walking distance, lower limb necrosis, claudication, and ischemic rest pain). Additionally, patients’ anthropometric measurements (height and weight values for body mass index [BMI] measured by the researcher) and their laboratory results were also obtained. Claudication was assessed by asking the following question, which refers to the definition of intermittent claudication: “Do you feel leg pain while walking which is relieved when your leg is at rest?” Patients were also asked whether they experienced resting leg pain and pain-free walking distance on a flat road. While evaluating the participants’ pain-free walking distance, two separate shopping centers were considered, 200 and 500 m away from the FHC, as reference points for accuracy of the distance. Patients’ last 6-month lipid profile, hemoglobin A1c (HbA1c), and fasting blood glucose values were obtained from the records of the FHC. Patients whose BMI was 30.0 kg/m2 or higher were considered as obese.
The ABI is a simple, inexpensive test to diagnose PAD [8]. For ABI calculations, classic Doppler method was used. Both left and right systolic blood pressures of the brachial, dorsalis pedis, and right tibial artery were measured while patients were resting in a supine position. Hand Doppler was used for accurate measurements of the lower limbs. ABI values were calculated by dividing the highest systolic pressure of the ankles by the highest systolic pressure of the arms [9]. ABI value between 0.90 and 1.30 was considered as normal, <0.9 as PAD, and >1.3 as noncompressible artery (NCA) [10,11]. Our study was approved by the University Medical Faculty Ethics Committee (approval no., 2011-01-05). A written informed consent for participation in the study was obtained from all participants.
Data were analyzed using the SPSS software ver. 11.5 (SPSS Inc., Chicago, IL, USA) package program. Besides descriptive analysis, chi-square test, Student t-test, and backward logistic regression analysis were used for comparative analysis. A P-value less than 0.05 was considered significant.
RESULTS
Descriptive characteristics of participants including PAD symptoms and ABI values are summarized in Table 1. The plasma lipid parameters of 224–226 patients were obtained from the laboratory data. Based on the measurements, 44 (17.6%) of participants had PAD, while 38 (15.2%) had NCA.
1. Peripheral Arterial Disease
Distribution of sociodemographic characteristics, risk factors, and PAD symptoms according to PAD status are shown in Table 2. BMI, renal disease, COPD, DM, family history of heart disease, CAD, smoking, and lipid, HbA1c, and fasting glucose levels were not statistically different among the participants with and without PAD (P>0.05). Of PAD (+) participants, 54.5% reported claudication-free walking distance over 500 m and 27.3% experienced less claudication. These two groups were considered as asymptomatic PAD.

Distribution of sociodemographic characteristics, risk factors, and symptoms according to presence and absence of PAD (n=250)
Women were at higher risk of PAD than men (P=0.020), but not in the regression analysis. Regression analysis revealed two predictors of PAD: age ≥65 years and claudication (Table 3).
2. Non-compressible Artery
The sociodemographic characteristics, risk factors, and PAD symptom distribution among the NCA (+) and NCA (-) participants were not statistically significant. Increased triglyceride (TG) values (P=0.010) and male sex (P=0.002) were significantly associated with NCA (Table 4). Among the participants with NCA, 15.8% walked a distance of less than 500 m, 39.5% experienced claudication, and 18.4% experienced ischemic rest pain. Meanwhile, logistic regression analysis showed that male sex, age <65 years, and TG >200 mg/dL were significantly associated with NCA ABI (Table 5).
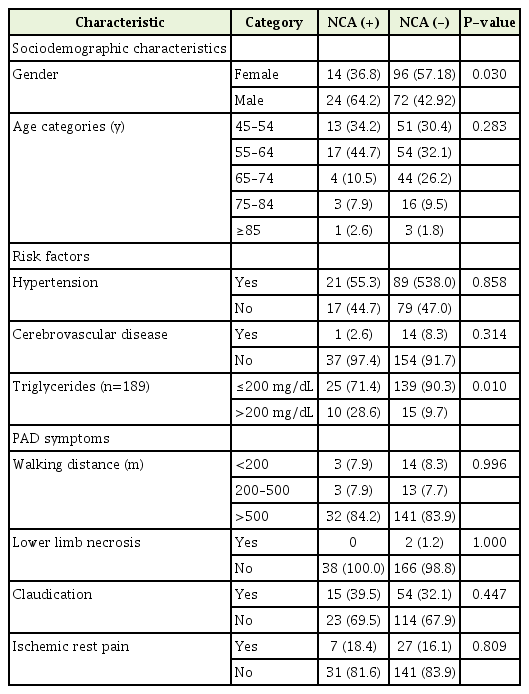
NCA distribution according to sociodemographic characteristics, risk factors, and PAD symptoms (n=206)
3. Sex Differences
Of the total participants, 142 were female patients (56.8%). The mean age of the female participants was 61.8±11.3 years old, while that of the male participants was 63.0±10.9 years old. With regard to sex, the values for PAD symptoms and PAD were higher in women (P<0.05) except lower limb necrosis (P=1.00). According to high total and LDL cholesterol levels, there was a statistically significant difference between men (36.8%; 34.7%) and women (58.8%; 51.9%) with p-values 0.001 and 0.008, respectively. On the contrary, no significant difference was observed between the values of TGs (P=0.692). Additionally, with a BMI cutoff point of 30 kg/m2, the prevalence of obesity was higher among women (49.3%) than men (23.1%) (P<0.001).
In this study, 22.4% of the participants were smokers. Half of the smokers were women. Smoking significantly decreased with age (≥65 years old) in both sexes (P<0.01).
DISCUSSION
1. Peripheral Arterial Disease
In our study, the prevalence of PAD was 17.6%, which was very similar to that reported in other studies in Turkey [6,7]. With regard to studies evaluating the prevalence of PAD, age was considered as an important risk factor and was found to have a positive correlation with the prevalence of PAD [12]. Moreover, the prevalence of PAD increased with age.
In our study showed that PAD was more prevalent in women (22.5%) than in men (11.1%). In terms of sex and prevalence of PAD, it can be inferred that men in general are at more of a disadvantage than women [13]. In one study, PAD was found in 3.9% of men and 3.3% of women, with a ratio of 1.18 [14]. Meanwhile, some studies showed no significant relationship between sex and prevalence of PAD [15,16]. Other previous studies showed that men had lower prevalence of PAD than women. In Rotterdam study, the prevalence of PAD was 16.9% among men and 20.5% among women, with a ratio of 0.82 [17]. However, a previous study indicated that although PAD is observed more frequently in elderly men, women more often develop this disease, regardless of age [18]. However, previous Turkish studies showed ambiguous sex-related results. In CAREFUL study, no significant difference was observed between sexes in terms of prevalence of low ABI. By contrast, another previous study showed the prevalence of PAD was higher in men than in women [6,7]. In our study, the total cholesterol levels, LDL levels, and BMI were higher among women; as these factors were considered as predictors of PAD, it may not be surprising that women have higher PAD prevalence than men [5,14].
Although a strong relationship between DM and low ABI was found in many studies, the association between DM and low ABI lost its significance after conducting multivariate adjustments in the Framingham Heart Study [1,2,14]. This association was not revealed in our study too. This may be due to the good blood glucose control among the diabetic participants. Considering the HbA1c values, there was no significant difference between PAD-positive participants and PAD negative participants. On the contrary, data on comorbid diseases were obtained by asking the participants and checking their medications. The fact that the data were obtained directly from the participants, the history may not be reliable, especially pertaining to kidney and heart diseases. This may also explain the absence of association between family history of heart disease and PAD.
Although smoking is a well-known risk factor for PAD, this association was not observed in our study.12,19) This may be due to the change of smoking prevalence among age groups. In our study, 91.1% of the smokers were aged <65 years old. We also found that age is an important risk factor of PAD. In the years to come, a predictable increase in PAD incidence may occur among smokers. In Turkey, there is a need for prospective and well-designed studies on this subject. However, since symptomatic patients with PAD have been advised to quit smoking, it is possible that observational comparisons may be confounded by other factors.
In the study, we evaluated the association between the PAD symptoms and PAD. The frequency of the symptoms among the PAD patients changes in terms of walking distance, necrosis, claudication, and rest pain. The presence of symptoms in patients diagnosed with PAD using ABI measurement strengthens the diagnosis. On the contrary, more than half of the PAD patients had walking distance more than 500 m, and one-third did not have claudication. These findings also support the diagnostic value of ABI measurement for asymptomatic PAD. These patients’ risks for coronary heart disease and prognosis were similar with those diagnosed with symptomatic PAD [20]. In 2010, previous study reported that >200 million people worldwide have PAD [21]. In this context, early diagnosis of PAD is important to prevent negative consequences. In our study, when walking distance was considered as the primary symptom, the asymptomatic PAD rate was 54.5%; when claudication was considered as primary symptom, the asymptomatic PAD rate was 27.3%. In existing literature, estimating the proportion of asymptomatic PAD remained a challenge [22]. Based on increasing physician and patient awareness, and the diagnosis of PAD in primary care, it has become possible to mention the asymptomatic prevalence of PAD patients.
2. Non-compressible Artery
Previous studies showed that an ABI value of >1.3 indicates non-elastic or calcified arteries [11]. Some investigators consider high ABI as an indicator of PAD since the proportion of PAD is high in this group [23]. Patients aged 50–64 years, with risk factors for atherosclerosis like hyperlipidemia, have increased risk of developing PAD [24]. In this study, among those with ABI values >1.3, age <65 years old, male sex, and higher serum TG levels (>200 mg/dL) were associated with high ABI values. Additionally, some patients with NCA had PAD symptoms like claudication and ischemic rest pain. A high ABI indicates poorly compressible infra-popliteal vessel, and this finding can mask the detection of PAD [25]. As 60%–65% of NCA patients have PAD, those with PAD symptoms should be referred to undergo imaging studies [26].
Conducting the study in a single FHC is one of the limitations of this study; however, as the study population was selected through random sampling, the results of the study can be applied in the general population and is the strength of the study. The samples, which only included the symptomatic patients without a comparison group or which only included those who visited the outpatient clinics, cannot show the risk and may have selection bias (the Berkson’s bias) [6,7]. Although our study population is small, the results of this study may serve as a basis for future meta-analysis. As our aim was to identify PAD patients using the ABI method in order to determine high-risk individuals, early diagnosis, optimum treatment, and referral should be performed among these patients in order to increase their quality of life.
In conclusion, our study showed that PAD diagnosed based on the ABI values was associated with age, sex, TG values, and claudication. While low ABI values (PAD) have been associated with age ≥65 years old and claudication, high ABI values (NCA) were associated with age <65 years old, TG >200 mg/dL, and male sex.
Although the identified PAD and NCA levels indicates the degree of PAD burden in local primary care, it is obvious that these characteristic differences among age groups need further research using a population-based study.
The reported number of patients with symptomatic PAD is just a ‘tip of an iceberg’; similarly, the number of asymptomatic patients remain unclear. Due to the silent nature of this disease, PAD has been considered the least effectively managed major atherosclerotic vascular disorder [27]. Almost half of all PAD patients seemed to have some clinical evidence of CAD or CVD, but only a small fraction of patients with PAD are properly diagnosed [28]. In this study, it is clear that when considering the rate of PAD, many asymptomatic PAD patients may be diagnosed only by implementing the ABI method in a more widespread manner.
Notes
No potential conflict of interest relevant to this article was reported.



