Effects of Galohgor Nutraceutical Lactation Cookies on Breast Milk Volume and Lactose Concentration
Article information
Abstract
Background
Galohgor nutraceutical lactation cookies (NLC) contain polyherbal which is believed and consumed by postpartum mothers of Sundanese Ethnic in Indonesia to increase breast milk production. This study was aimed to analyze the effects of NLC cookies on breast milk volume and lactose concentrations of postpartum mothers.
Methods
The experimental study with post-controlled design was conducted. Twenty eligible mother-infant pairs were enrolled and divided into two groups. They were randomly assigned to receive NLC (n=9) or original cookies (ORC, n=11). Each subjects consumed 4 pieces/d of NLC which containing 1 g galohgor nutraceutical powder/ pieces or ORC (without galohgor nutraceutical powder), for 14 days intervention period since the first day of giving birth. The measurement of breast milk production was carried out by baby weighing method using SECA 334 digital baby scale (SECA, Hamburg, Germany). A high-performance liquid chromatography method was used to determine lactose concentrations in breast milk. Data were analyzed by parametric Independent T-test or the nonparametric Mann-Whitney U-test and adjusted analysis of covariance.
Results
NLC significantly increased (P<0.05) breast milk production, as shown by average breast milk volume of NLC compared to ORC, i.e., 557.0 (95% confidence interval [CI], 497.3–616.6) mL/d vs. 435.9 (95% CI, 382.3–489.6) mL/d. This result was supported by lactose concentration in breast milk of mothers who consumed NLC significantly higher (P<0.05), compared to ORC, i.e., 6.03% (95% CI, 5.66%–6.40%) vs. 5.18% (95% CI, 4.85%–5.51%).
Conclusion
NLC which containing galohgor nutraceutical can be recommended to increase breast milk volume and lactose concentration in the early postpartum period.
INTRODUCTION
The World Health Organization has strongly recommend exclusive breast feeding for 6 months of early life in order to give adequate nutrients of infants, and continued by complementary feeding that should be added by breast milk for 2 years [1]. Breast milk has been accepted as the gold standard of infant nutrition. Adequate breast milk production of mother is very essential for the successful of breast feeding. Early breast milk production has been shown to significantly affect breast milk production during established lactation period [2].
Many cultures believe that certain food increase breast milk production during breastfeeding. Galohgor nutraceutical is a traditional nutraceutical made of 56 kinds of plants that contains β-carotene, bioactive compounds, and other various nutrients. This nutraceutical is usually consumed by Sundanese Villager in Indonesia to increase milk production, to promote uterine involution and health status of postpartum mothers [3,4].
Our preclinical study showed that galohgor nutraceutical which contain β-carotene induce differentiation and function of the mammary gland cell line (HC11) [5]. Meanwhile, several studies showed that β-carotene increase connexin (Cx43) gene expression and gap junction intracellular communication (GJIC) which leads to cell differentiation [6,7]. GJIC is a channel like structure in the cell membrane that is developed by connexin that maintain cell membrane permeability, regulates cell metabolism, and differentiation. During lactation, the development of the mammary gland GJIC is very critical for sustainability of breast milk production [8,9].
Practically, the total volume as amount of milk production is a key element defining lactation performance. Lactose, a β-disaccharide consisting of glucose and galactose, is the most principal carbohydrate in human milk (90%–95%). Of all mammals, lactose is present in the highest concentration in the milk of humans (approximately 7 g 100 mL-1), corresponding to the high energy demands of the human brain [10]. Many previous studies indicated that the rate of lactose synthesis is a major controlling factor of breast milk production [11].
Understanding volume and lactose concentration of breast milk provide an important tool for management of infant feeding. Meanwhile, the effect of nutraceutical lactation cookies on breast milk production and lactose concentration of breast milk has not been studied. The objective of this study was to evaluate the effect of nutraceutical lactation cookies effects on breast milk production and lactose concentration of breast milk in early postpartum period.
METHODS
1. Subjects and Study Design
This study was experimental study with post-controlled design. The study protocol was approved by Committee of Human Rights Related to Research Involving Human Subjects, IPB University, Indonesia (No.081/IT3.KEPMSM-IPB/SK/2018). All subjects were informed and gave their consent before enrollment.
This study was conducted at Primary Health Care Center, Bogor City, West Java Province, Indonesia. A total of 24 registered breastfeeding mother-infant pairs were screened, 20 were enrolled. The subjects included aged between 26 to 40 years, single pregnancy, normal delivery, multiparous, having willingness to exclusively breastfeed their infants, and having consented to follow-up visits.
Subjects were excluded if they were having any medical condition or complications during previous pregnancies or delivery, having breast problems such as inverted nipples and mastitis and using a medicine, smoking, drinking alcohol, and being separated from their infant. All infants enrolled into the study were term and healthy neonates. Infants with low birth weight, intrauterine growth retardation and any illness or congenital abnormalities were excluded. After the initial screening visit, 20 eligible subjects were randomly assigned to receive galohgor nutraceutical lactation cookies (NLC, n=9) or original cookies (ORC, n=11) (Figure 1). Randomization was done according to computer-generated random number sequence. Cookies intervention groups were allocated by research assistant. Participants and principal investigators were blinded to group allocation.
2. Intervention Procedure
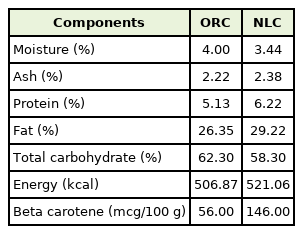
The subjects received 4 pieces (40 g) per day of NLC or ORC. NLC contained 1 g galohgor nutraceutical powder per pieces. The galohgor nutraceuticals powder has been already registered on Indonesian patent number IDP000058958 [12]. NLC were made by CV Nutrasetikal Galohgor (NutriLaktasi, Jakarta, Indonesia) using a cookies formulation that has been acknowledged as a household food production unit. ORC was manufactured without galohgor nutraceutical powder and matched to the NLC in size, shape, and color (as placebo). The nutrient composition of the NLC and ORC cookies is presented in Table 1. All subjects took the assigned cookies for 2 weeks intervention period starting on the first day of giving birth. They were asked to maintain their usual food intake throughout intervention study. We also investigated subject’s diary administration and cookies count (both NLC and ORC) every visit to monitor compliance.
3. Characteristics of Subjects
We collected maternal and infant characteristics data including maternal age, parity, gestational age, education, socio-economic status (family size and wealth status), and gender of infants by interview using questionnaires at the initial (week 0) visits (Figure 1). Anthropo- metric measurements of mothers were obtained through a physical examination on day 13. Height was measured to the nearest precision of 1 mm by using a stature meter whilst body weight was measured with minimum clothing to the nearest precision of 0.1 kg by using a Camry digital weighing scale (Camry Co., Zhongshan, China). Body mass index (BMI, kg/m2) was calculated to determine nutritional status of the mothers. Body fat percentage was measured using bioelectrical impedance analyzer (Omron Co., Kyoto, Japan). The maternal prolactin level was measured on the second week of postpartum period (day 14) in Prodia Laboratory, Bogor, Indonesia. A fasting blood sample was drawn by a research nurse during 8–9 AM of the visit day. Infant anthropometric data (weight, length, and head circumference) were obtained on day 14. Weight and length of infant were measured by using digital baby scale (SECA 334 and SECA 232; SECA, Hamburg, Germany) meanwhile head circumference was measured by using standard measuring tape.
4. Dietary Assessment
Dietary intake was assessed by using 24 hours recall method, conducted twice for each subject for nonconsecutive days on week 1 (Figure 1). We determined the results of the two evaluations by calculating the mean of dietary intake from two non-consecutive 24 hours recall during a time span of a week. The food recalls provided information on portion size using household measures and quantities (g). Dietary intake was calculated with the nutrient content of the consumed intervention cookies. Calculation of energy and macronutrient intake was based on Indonesian Food Composition Database. Macronutrient intake was analyzed using Nutrisurvey 2007 software package downloaded from www.nutrisurvey.de.
5. 24-Hours Breast Milk Volume Measurement
Breast milk intake measurement were based on Brown et al. [13] In test weighing, infants are typically weighed before and after each feeding without a change of clothing or diapers during 24-hour period recorded. The difference in body weight (in grams) is considered to approximate the volume of milk consumed (in mL) after multiplying by 0.983 mL/g to adjust for the density of breast milk. The weight of the infants was measured using a baby electronic weighing scale accurate to ±10 g (SECA 334; SECA). The scale was periodically checked for its accuracy by a calibration weight.
6. Breast Milk Sample Collection
Breast milk samples were collected on day 14 for lactose concentration analysis apart form 24-hour breast milk volume measurement. A milk sample of 50 mL was obtained in the morning between 8 to 9 AM for each mother by electrical breast pump (Spectra S1; Uzinmedicare Inc., Hwaseong, Korea). The samples were collected in a sterile bottle, transported on ice (4°C) to the laboratory unit and stored at -20°C in local freezers until analysis.
7. Lactose Concentration Analysis
1) Preparation of Standards
A high-performance liquid chromatography (HPLC) method was used to determine lactose concentration in breast milk (AOAC [Association of Official Analytical Collaboration] Official Method 980.13) [14]. Lactose standard was obtained from Sigma-Aldrich Chemical Co. (St. Louis, MO, USA). Standard solution of lactose at a concentration range between 0.005% and 1% was prepared and used to create the calibration curve. Prior to injection, all standards were filtered into individual 2 mL vials through 0.45 µm filters to remove any small particles.
2) Sample preparation
In this study, 2.5 g of breast milk sample was weighed into centrifuge bottle and 25 mL petroleum ether was added. Breast milk sample was centrifuged 15 minutes at 1,800 rpm. The supernatant was decanted and discard into a clean test tube. Then, the extraction was repeated 3 times. Afterward, the residue was pulverized with glass rod, 100 g H2O was added and the residue was weighed. The residue was placed in 85°C–90°C H2O bath 25 minutes and then cooled to room temperature. H2O was added to original weight. Then, the residue was centrifuged 10 minutes at 2,000 rpm with draw portion of clear supernatant and filtered into individual 2 mL vials using 0.45 µm filters. After that, 10 µL of prepared sample was then injected onto the HPLC.
8. Statistical Analysis
Difference of quantitative variables, according to their distribution, was analyzed with the parametric Independent T-test or the non-parametric Mann-Whitney U-test. To account for the potential effect of galohgor cookies on final outcome variables of the 14 days lactation period were estimated by analysis of covariance analysis. The results of outcome variable, i.e., breast milk volume and lactose concentration are reported as adjusted mean value and lower-upper limit of 95% confidence intervals (CI). Meanwhile, the subject’s characteristics and dietary intake are reported as mean values and standard deviation. In all statistical comparisons, differences with P<0.05 were considered significant.
RESULTS
1. Subjects Characteristic
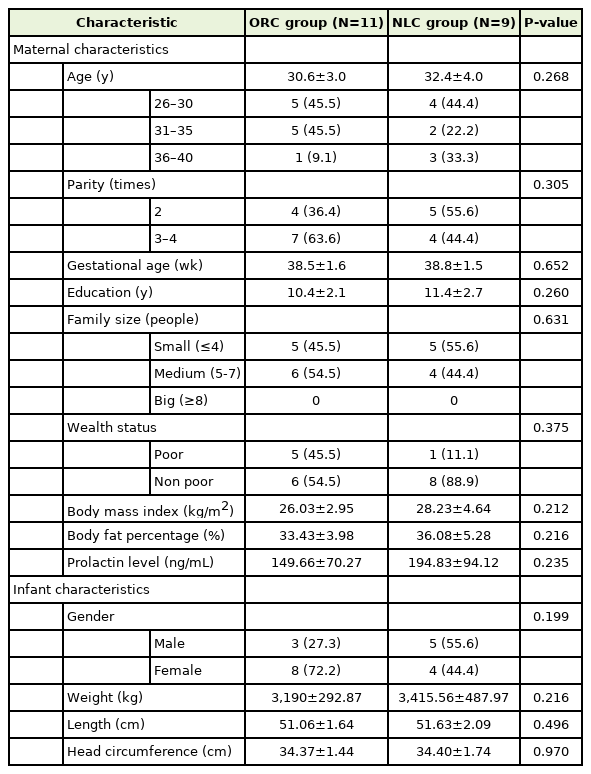
Characteristics of mother-infant pairs are elaborated in Table 2. Maternal age, parity, gestational age, education, family size, wealth status, BMI, and body fat percentage, and prolactin concentrations were not significantly different between two groups. Furthermore, infant characteristics such as gender, weight, length, and head circumference of these two groups are also not significantly different.
2. Macronutrient Intake
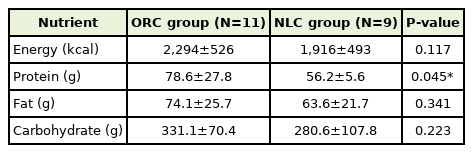
Table 3 shows estimated dietary intake of mothers during intervention period. Concerning the intake value of energy, fat, and carbohydrate, no significant difference between the two groups were observed. Meanwhile, the intake value of protein was significantly different in both groups.
3. Breast Milk Production
The 24-hours breast milk volume were measured on day 14 postpartum and the results are shown in Figure 2. The result shows that the mean of breast milk volume in intervention group of NLC is significantly higher than ORC (557.0 mL/d [95% CI, 497.3–616.6 mL/d] versus 435.9 mL/d [95% CI, 382.3–489.6 mL/d]) after being adjusted for parity, gestational age and head circumference of infants.
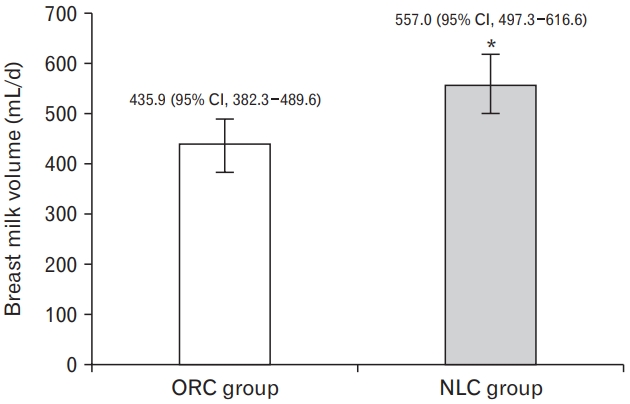
Breast milk volume of the intervention groups. Adjusted mean value and lower-upper limits of 95% confidence interval (CI) error bars are presented. Intervention effect was estimated by using adjusted analysis of covariance. The covariates are parity, gestational age, and head circumference of infant. NLC, Galohgor nutraceutical lactation cookies. *P<0.05; significantly higher than original cookies (ORC) group.
4. Lactose Concentration of Breast Milk
As shown on Figure 3, the mean concentration of lactose in intervention group of NLC is found significantly higher than ORC (6.03% [95% CI, 5.66%–6.40%] versus 5.18% [95% CI, 4.85%–5.51%]) after being adjusted for parity, carbohydrate intake of mother and water content of breast milk.
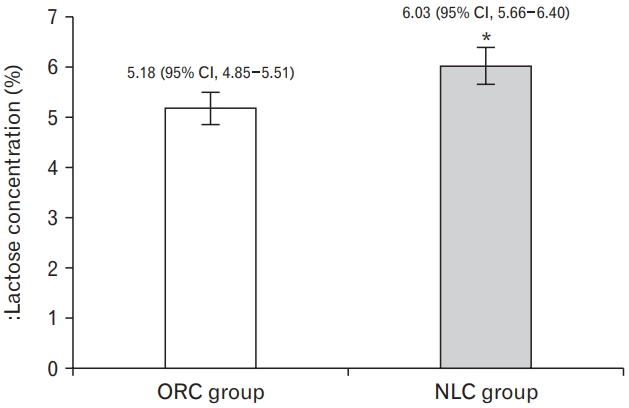
Lactose concentration of the intervention groups. Adjusted mean value and lower-upper limits of 95% confidence interval (CI) error bars are presented. Intervention effect was estimated by using adjusted analysis of covariance. The covariates are parity, water content of breast milk, and carbohydrate intake of mother. NLC, Galohgor nutraceutical lactation cookies. *P<0.05; significantly higher than original cookies (ORC) group.
DISCUSSION
Breast milk volume measurement is the standard method to assess the performance of breast milk production. Our finding shows that the intervention of NLC significantly affecting on higher breast milk production of NLC compared to ORC (557.0 mL/d [95% CI, 497.3–616.6 mL/d] versus 435.9 mL/d [95% CI, 382.3–489.6 mL/d]; P<0.05). In accordance with our previous animal study, the milk production of galohgor groups were significantly higher than control groups on day 7 of lactation period of rats [15].
The 24-hours breast milk production of NLC group was within the normal range for established lactation (440 to 1,220 mL/d) during the second week after birth [16]. The average of breast milk production NLC group (557.0 mL/d [95% CI, 497.3–616.6 mL/d]) in this study result is higher compared to the data of galactagogue intervention presented by several studies. In the study of Swafford and Berens [17], 10 mothers consumed three cups of fenugreek daily for 2 weeks intervention. Their results showed that breast milk production increased from 207 mL/d in the first week to 464 mL/d in the second week. Damanik et al. [18] revealed that during the last 2 weeks of Coleus amboinicus or “torbangun” supplementation (from day 14 to day 28), the 24-hour breast milk volume was significantly increased from 361 to 479 mL, or with the average increase of 65%. In addition, the differences between results of previous studies with our present study may be due to the type of nutraceuticals or galactagogue, the dosage, and intervention periods. However, the method of the calculating breast milk production was similar.
The mechanism of galohgor effect on increasing breast milk production has been elaborated by several studies that compromised each other. According to Roosita et al. [5] and Ma’rifah et al. [19] β-carotene is the most carotenoid compound present in galohgor nutraceuticals. Our previous in vitro study showed β-carotene plays an essential role in the physiological function of mammary gland differentiation [5]. Galohgor influenced aldehyde dehydrogenase (ALDH1A2) gene expression which plays important roles in the metabolism of β-carotene [20]. β-carotene is absorbed in the small intestine in the form of retinol and transported in blood attached to retinol-binding protein. Inside the cells, retinol is oxidized into its main biologically active derivatives, retinaldehyde (retinal) and retinoic acid. After conversion of β-carotene to retinal which is catalyzed by β-carotene-15-15-dioxygenase, ALDHs catalyzes retinal oxidation to retinoic acid [21,22]. Retinoic acid is an important regulator of cell differentiation and plays a major role in embryonic development and tissue remodeling. The biological action of retinoic acid is mediated by three nuclear receptors, RARα, β, and γ, which are ubiquitously expressed in the breast. Moreover, several studies have shown that retinoic acid signaling is required for development of the mammary gland after birth [23].
Furthermore, our previous study showed that β-carotene increased mammosphere development, genes expression of connexin (Cx43) and β-casein (Csn2) of the mammary gland cell line (HC11). Connexin is a protein that develops gap junction structure in neighboring mammary epithelial cell, meanwhile casein (major milk protein) gene expression is an important marker for lactogenesis [5]. Thus, β-carotene is an important compound that explains the mechanism of nutraceutical galohgor effect on mammary gland differentiation. An appropriate dietary intake of β-carotene during lactation together with the inter tissue flux of different nutrients in the gland during lactation influence vitamin A concentration in breast milk [23]. Galohgor nutraceutical also have beneficial effects on health status of breastfeeding mothers by improving β-carotene serum levels and oxidative stress [19]. Tanaka et al. [24] suggested that oxidative stress, a consequence of redox imbalance, has been associated quantity and quality of milk.
Breast milk composition varies considerably within and between mothers and even within a single milk expression.
Our finding shows that consumption of NLC during early postpartum is significantly affecting on higher lactose concentration on breast milk compared to ORC (6.03% [95% CI, 5.66%–6.40%] versus 5.18% [95% CI, 4.85%–5.51%; P<0.05) on day 14th of lactation. This result is also comparable with two studies of lactose concentration on breast milk which collected from day 11th to 28th of lactation was 6.50% [25], whilst for 2 months of lactation was 6.53%±0.34% [26]. According to Arthur et al. [11] high concentration of lactose in milk implies that lactogenesis II has occurred and milk synthesis tends to be higher.
Lactose concentration was influenced by lactation period, breastfeeding pattern, and mother’s or offspring’s characteristics [10]. Lactose production is the highest in the fourth to 7 month, after which it decreases, whilst a gradual increase in the lipid concentration occurs over lactation [27]. However, Aumeistere and Zavadska [26] revealed that lactose concentration in mature milk is stable and insensitive to changes. According to Khan et al. [28] during the established lactation, higher concentration of lactose in breast milk is associated with higher 24-hour breast milk volumes and higher breastfeeding frequency. Meanwhile Dewey et al. [29] and Verd et al. [30] revealed that when breast milk volume felt down below 200 mL/d, the lactose, calcium, and zinc decreased whereas protein and sodium increased.
The study showed that the adverse effects were not found in both mothers and infants. The researchers interviewed the mothers regarding the adverse effects of NLC and ORC, including headache, rash, itching, nausea, abdominal pain, and excessive rectal gas. Some previous study reported that galohgor is safe for consumption based on survey in community [3].
The differences between the NLC and ORC groups in the changes of lactose concentration as the results of galohgor effect was compromised with the enhancement of breast milk production. Nevertheless, there were some limitations of this study; this study has a small sample and had a short study duration of only 2 weeks. The strength point of this study was the new finding of galohgor nutraceutical cookies administration that provide positive effects in breast milk volume and lactose concentration of postpartum mothers in Indonesia. This study also used a 24-hour test weighing method, which is considered as the gold standard for measuring breast milk volume. In addition to the use of galohgor as nutraceutical, the importance of lactation counseling should be never ignored.
In conclusion, NLC can be recommended as traditional nutraceutical to increase breast milk volume and lactose concentrations in the early postpartum period.
Notes
CONFLICT OF INTEREST
No potential conflict of interest relevant to this article was reported.
Acknowledgements
This study was financially supported by Ministry of Research, Technology and Higher Education of Republic Indonesia through LPPM, IPB University, for which we would like to express our gratitude and appreciation. We are also grateful to Almira Nuraelah and Humairoh Sholehah for their assistance in the data collection. The authors also greatly appreciate the kind cooperation from Public Health Office of Bogor City, the midwifes and nurses at primary health care center of Kayumanis, Cipaku, Pancasan, and Warung Jambu in Bogor City, cadres, and subjects in the studied communities.