Short-term Changes in Thyroid-Stimulating Hormone Level after Body Fat Reduction via Partial Meal Replacement
Article information
Abstract
Background
Serum thyroid-stimulating hormone (TSH) levels change during body weight reduction. However, the changes that occur during short-term body weight control interventions remain controversial. Thus, this study aimed to evaluate the changes in TSH levels according to body fat reduction.
Methods
We performed a 3-month intervention study involving partial meal replacement. Forty-nine participants completed the study. Correlations between changes in TSH levels and other body composition parameters were determined. The subjects were divided into two groups according to their body fat reduction (>1 kg, n=20; <1 kg, n=29). The changes in metabolic parameters, including TSH levels, were compared. For significant values, a multivariate analysis was performed after adjustment to evaluate the relationship between TSH changes and body fat reduction.
Results
The 3-month intervention caused favorable changes in body proportions and metabolic parameters. TSH levels changed significantly only after changes in total body fat, showing a partial correlation. Changes in TSH levels were significantly different between groups (P=0.014). Moreover, the change in TSH levels was significantly different after adjustment (P=0.012).
Conclusion
A body fat reduction, especially >1 kg, can reduce serum TSH concentrations in subjects with metabolic syndrome after short-term body weight intervention.
INTRODUCTION
Metabolic syndrome is a well-known cardiovascular disease. Central obesity, one major factor of metabolic syndrome, is reportedly closely associated with cardiovascular morbidity and mortality [1]. Therefore, a reduction in body weight via exercise and diet control is the main strategy to prevent cardiovascular diseases in patients with metabolic syndrome. A previous study showed that meal replacement with mixed rice controls weight and improves the antioxidant enzyme activities of obese women [2]. We also reported that black rice with giant embryos can improve metabolic derangement through weight control and reducing vascular inflammation [3]. Therefore, the long-term use of partial meal replacement with rice powder may effectively reduce fat mass and improve various metabolic parameters.
Levels of thyroid hormones can change during the weight control period. Thyroid hormones play important roles in the regulation of lipid metabolism. Only a slight change in serum thyroid hormone concentration can cause local fat accumulation and increase body mass [4,5]. Recent studies reported that thyroid-stimulating hormone (TSH) can be directly blinded to the TSH receptor in tissues outside the thyroid (such as the adipose tissue), thereby having external effects [6]. However, most studies of the relationship between thyroid hormone parameters are derived from drastic interventions, such as bariatric surgery or the manipulation of macronutrient composition [7-9], while limited information is available about thyroid hormone metabolism during weight loss achieved by partial meal replacement. Moreover, changes in TSH levels can be interpreted as an adaptation to a reduced fat content. After adaptation, the total body energy expenditure is reduced to some extent. This might be a cause of difficulty maintaining a reduced body weight. Therefore, this study aimed to explore the changes in TSH levels in body weight reduction subjects with metabolic syndrome, especially in the fat reduction group, during a 3-month weight control program consisting of partial meal replacement.
METHODS
1. Study Design and Subjects
These methods were described in our previous study in which we evaluated changes in metabolic biomarkers after partial meal replacement (black rice or white rice) for 3 months [3]. Subjects aged 20–59 years with a body mass index of 25–30 kg/m2 were included. Subjects received 1 month of meal supplements consisting of black rice with a giant embryo or white rice instead of breakfast at every monthly visit. The meal replacement was provided in the same colored disposable small plastic bag containing black rice with giant embryos or white rice. The participants were asked to maintain their usual exercise and food intake.
Subjects who had a black rice allergy, type 2 diabetes, knee osteoarthritis, chronic obstructive lung disease, tuberculosis, hypothyroidism, or recent cardiovascular diseases (myocardial infarction and variant angina); who were currently pregnant or breastfeeding; or had psychological problems or cancers were excluded. Patients taking various steroids were also excluded from the study.
Fifty-one participants with metabolic syndrome were recruited for this study. Two dropped out due to moving to other cities. Finally, 49 subjects finished the study. Subjects who had metabolic syndrome and were >20 years of age participated in this study after providing voluntary consent. In the present study, we did not divide the subjects into black and white rice groups. Instead, we divided the subjects into two groups according to the amount of body fat loss (fat reduction >1 kg, n=20; fat reduction <1 kg, n=29) to compare TSH levels between them. The Institutional Review Board of Ajou University Hospital approved this study (approval no., AJIRB-MED-CT3-15-248) after obtaining proper informed consent.
2. Measurements
Metabolic parameters such as fasting glucose, insulin, lipid (Toshiba 200FR automatic analyzer; Toshiba Medical Systems, Tokyo, Japan), cortisol, high-sensitivity C-reactive protein (hs-CRP; Dade Behring Marburg GMBH, Marburg, Germany), and thyroid hormone (TSH and free T4) levels were assessed, while body proportions were measured by dual-energy X-ray absorptiometry (DISCOVERY-W fan-beam densitometry; Hologic Inc., Marlborough, MA, USA) before and after the intervention (at baseline and 3 months). Body weight, waist circumference, blood pressure (standard mercury manometer with the participant in a sitting position for 5 minutes prior to measurement), and adverse effects were also evaluated by a registered research nurse at every visit. Total calorie intake was assessed using a self-administered 24-hour food diary by a trained dietitian monthly. Metabolic syndrome (National Cholesterol Education Program Adult Treatment Panel III) was defined as the presence of three or more of the following risk determinants: (1) central obesity (waist circumference: ≥90 cm in men, ≥85 cm in women); (2) high fasting glucose level (≥126 mg/dL); (3) high blood pressure (≥130/85 mm Hg) at rest; (4) high triglyceride level (≥150 mg/dL); and (5) low high-density lipoprotein cholesterol (≤40 mg/dL in men, ≤50 mg/dL in women). Blood tests were performed after overnight 8-hour fast.
3. Statistics
All values are presented as mean±standard deviation. Data from all study subjects before and after the intervention were compared using a paired t-test. Correlations between changes in body composition and TSH levels were also evaluated by partial correlation after the adjustment for age, sex, smoking, alcohol intake, and moderate-intensity exercise. Subjects were divided into a >1 kg body fat reduction group and a <1 kg body fat reduction group. In this study, the number of participants was not large enough to be categorized. Therefore, the cutoff value for body fat reduction was determined as 1 kg. The mean changes in metabolic parameters were compared between groups. We also compared significant values after adjusting for age, sex, smoking, alcohol intake, and moderate-intensity exercise using a general linear model. Statistical significance was set at P<0.05. The data were analyzed using IBM SPSS ver. 19.0 (IBM Corp., Armonk, NY, USA).
RESULTS
The anthropometric and metabolic parameters of the study subjects at baseline and 3 months after the intervention are summarized in Table 1. As predicted, the subjects had elevated triglyceride levels and waist circumferences at baseline. After the 3-month intervention, body weight, waist circumference, total fat mass, diastolic pressure, and hs-CRP levels were significantly reduced. However, TSH and free T4 concentrations showed no significant changes.
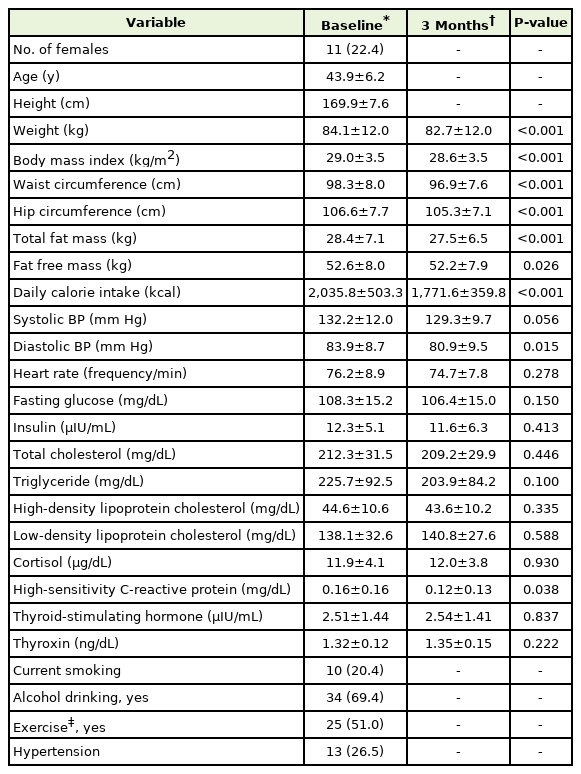
Anthropometric and metabolic parameters of study subjects (n=49) at baseline versus 3 months after intervention
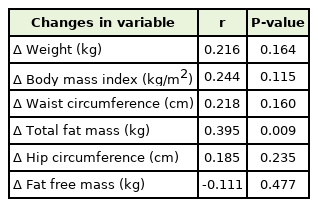
Changes in TSH levels showed a positive correlation with total fat changes after adjustment (P=0.009) (Figure 1). The partial correlations of TSH changes with body composition are shown in Table 2. TSH was significantly correlated with changes in total fat mass (P=0.009) after the adjustment for age, sex, smoking, alcohol consumption, exercise, and hypertension.
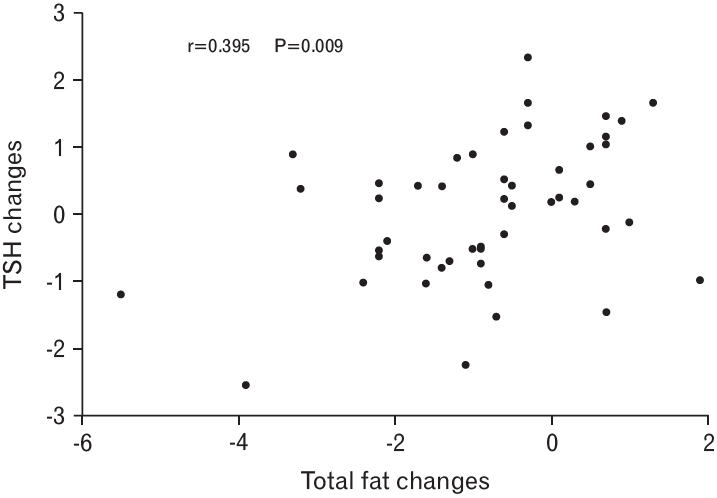
Correlations between total body fat changes and thyroid-stimulating hormone (TSH) changes after adjustment.
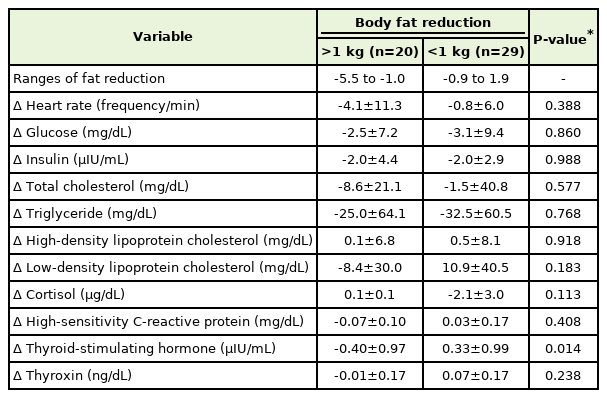
We divided the subjects into two groups according to the amount of fat reduction and investigated the changes in metabolic parameters (Table 3). Changes in serum TSH concentrations in the group with a fat reduction of >1 kg showed a significant decrease (P=0.014). After the adjustment for age, sex, smoking, alcohol consumption, and moderate-intensity exercise, TSH still showed a significant decrease in the group with a fat reduction of >1 kg (P=0.012) (Table 4).
DISCUSSION
The present study showed a significant decrease in serum TSH concentration among subjects with metabolic syndrome who experienced a body fat reduction of >1 kg after a 3-month intervention of partial meal replacement with rice powder. The relationship between thyroid hormone levels and body fat mass remains unclear. In an interventional trial of 47 overweight subjects, TSH and triiodothyronine levels were significantly associated with fat mass at baseline [10]. In a crosssectional study of euthyroid adults with known vascular disease, higher serum TSH levels were associated with increased visceral fat thickness among individuals aged 67–80 years [11]. A previous study of 303 healthy volunteers demonstrated that the amount of subcutaneous fat was positively correlated with TSH level [12]. However, conflicting results have also been reported. A prospective study using meal supplements showed no significant difference in TSH levels between the body fat reduction group and other groups [13]. A controlled follow-up study reported that thyroxine treatment in patients with hypothyroidism increased the resting energy expenditure and decreased body weight, whereas fat mass was unchanged with weight loss, primarily due to the excretion of excess body water [14].
It is well established that thyroid hormone status is correlated with energy expenditure [15]. Thyroid hormones can stimulate both lipogenesis and lipolysis, although the net effect is fat loss when thyroid hormone levels are elevated [16]. Fatty acids produced from thyroid hormone– induced lipolysis are substrates for increased thermogenesis [17]. Bile acid–stimulating pathways include direct actions on cholesterol metabolism. They can also stimulate 5-deiodinase type 2 activity and thyroid hormone–mediated increases in energy expenditure. Although the reason for the decrease in TSH levels among those with decreased body fat is not entirely clear, we assume that it is an adaptive process that decreases energy expenditure and encourages fat accumulation [18]. Leptin may also play a role in regulating TSH and fat mass. Leptin is known to regulate the production of thyrotropin-releasing hormone in neurons within the periventricular nucleus [19,20]. Moreover, leptin can activate thyrotropin-releasing hormone neurons via Janus kinase/signal transducers and transcription pathway activators [20,21]. An increase in thyrotropin-releasing hormone levels can lead to a higher pituitary secretion of TSH [21-23].
The effects of body weight reductions on thyroid status were recently investigated. Among 783 obese patients who underwent a bariatric surgery evaluation, 18.1% had elevated serum TSH levels [24]. In a crosssectional analysis of data from the National Health and Nutrition Examination Survey, among 3,114 euthyroid adults without a history of thyroid disease, body mass index and waist circumference were positively associated with serum TSH levels [25]. Some studies have reported that weight loss can lead to normalized serum TSH levels [24]. However, serum TSH concentration made no difference between before and after meal replacement in our study. This may be related to the concept of the lipostatic setpoint as a time interval for weight regulation. We found that TSH levels could change within normal ranges with reductions in body fat. Although long-term intervention trials are needed, short-term reductions in body fat are associated with TSH changes, which can be related to total body fat or body weight reductions. Therefore, clinicians should recognize TSH changes according to changes in body fat as well as metabolic parameters.
This study had some limitations. First, it evaluated only short-term changes. Second, our data analysis involved a small number of subjects other than those in the conventional diet program. Thus, the results of our study should be confirmed in larger trials.
In conclusion, body fat reductions, especially those >1 kg, can change the serum TSH concentrations of subjects with metabolic syndrome. Long-term intervention studies are warranted to validate these findings.
Notes
CONFLICT OF INTEREST
No potential conflict of interest relevant to this article was reported.
Acknowledgements
This study was funded by the National Institute of Crop Science, Rural Development Administration of the Republic of Korea.