 |
 |
- Search
| Korean J Fam Med > Volume 41(3); 2020 > Article |
|
Abstract
ACKNOWLEDGMENTS
Figure. 1.
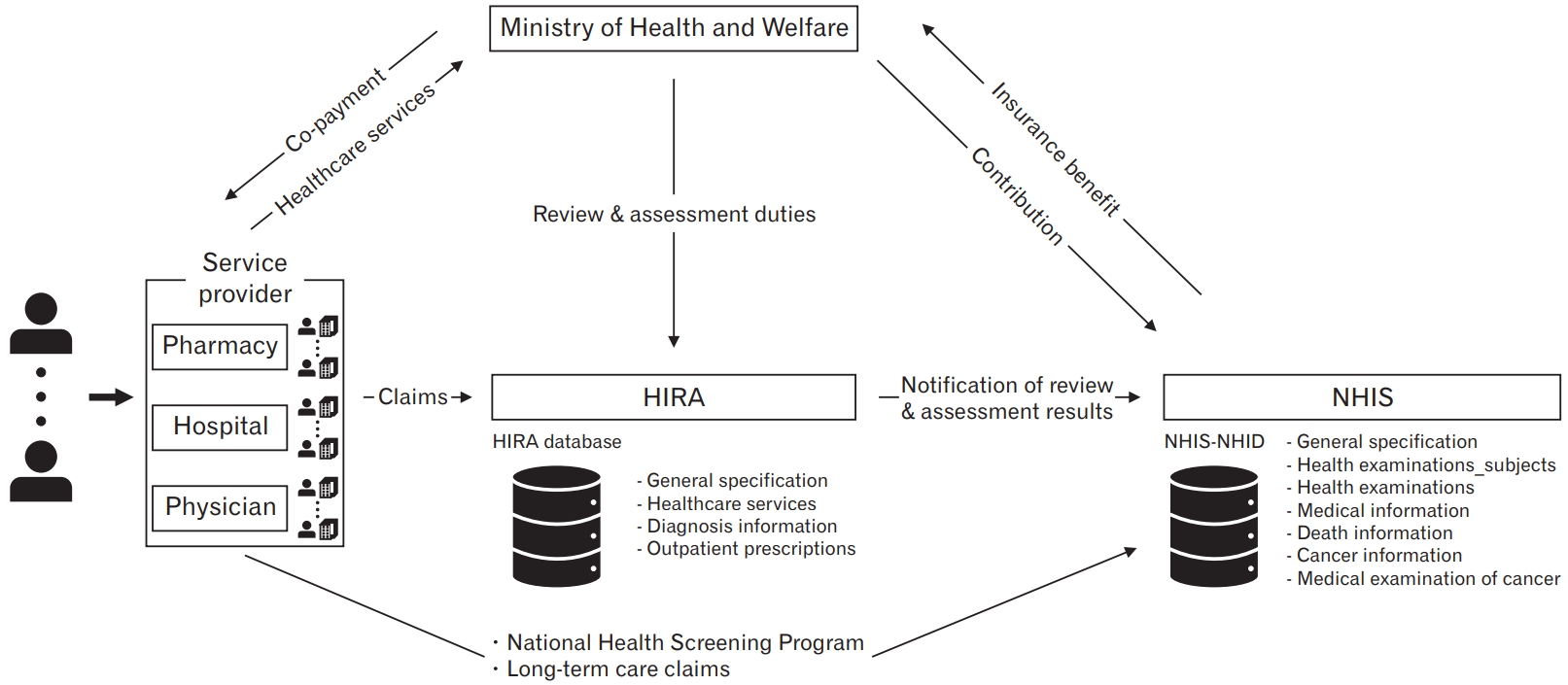
Table 1.
HIRA, Health Insurance Review and Assessment Service; HIRA-NPS, HIRA-National Patient Sample; HIRA-NIS, HIRA-National Inpatient Sample; HIRA-APS, HIRA-Aged Population Sample; HIRA-PPS, HIRA-Pediatric Patient Sample; NHIS, National Health Insurance Service; NHIS-NHID, NHIS-National Health Information Database; NHIS-NSC, NHIS-National Sample Cohort; NHIS-HEALS, NHIS-National Health Screening Cohort; NHIS-FEM, NHIS-Female Employees; NHIS-INCHS, NHIS-Infants and Children’s Health Screening.
Table 2.
| Source | Database | Data period | Contents and variables* |
|---|---|---|---|
| HIRA | HIRA database | 2007–2018 | - General specifications (billing statement identification key, age, gender, type of insurance, date of treatment, primary diagnosis, secondary diagnosis, surgery, etc.) |
| - Healthcare services (billing statement identification key, inpatient prescriptions, treatments, diagnostic tests, unit price, days of supply, etc.) | |||
| - Diagnosis (billing statement identification key, diagnostic code, department, etc.) | |||
| - Outpatient prescriptions (billing statement identification key, drug codes, unit price, days of supply, etc.) | |||
| NHIS | NHIS-NHID | 2007–2018 | - General specifications (year, age, gender, region, grade of disability, contribution amount, etc.) |
| - Health examinations - subjects (year, working type) | |||
| - Health examinations (disease history, physical activity, current medications, smoking, drinking, height, weight, blood pressure, laboratory tests, etc.) | |||
| - Medical institution (year, location, number of doctors, number of nurses, number of pharmacists, number of beds, etc.) | |||
| - Death information (death year and month) | |||
| - Cancer information (breast/colorectal/cervical/liver/gastric cancer) | |||
| - Medical examination of cancer (medical examination experience, medical history, year, family history, etc.) | |||
| NCC | KNCI DB | 2002–2016 | - Age, gender, date of diagnosis, Surveillance Epidemiology and End Results code, diagnosis code, primary cancer site, treatment, histological type, etc. |
| KCDC | KNHANES | 2007–2017 | - Age, gender, socioeconomic status, educational status, chronic disease, health status, cancer examination, cost, quality of life information, injury, height, weight, blood pressure, laboratory tests, nutritional intake, dietary supplements, nutritional knowledge, etc.) |
| KCDC | Quarantine database | 2013–2018 | - Date of quarantine, type of quarantine, site of quarantine, country of departure, transportation, number of crew, number of passengers, number of suspicious entrants, pollution, major freight |
| KCDC | KTBS system database | 2013–2018 | - Year, age, age group, gender, region, nationality, reporting public health center, reporting medical institution, date of reporting, type of tuberculosis, disease code, patient type, smear screening |
| KCDC | KoGES | 2001–2013 | - Cohort name, age, gender, chronic disease, smoking, drinking, exercise, blood pressure, height, weight, laboratory tests, etc. |
| KCDC | Immunization registry data | 2012–2018 (only NIP) | - Vaccination name, date of vaccination, medical institution, region of medical institution |
HIRA, Health Insurance Review and Assessment Service; NHIS, National Health Insurance Service; NHIS-NHID, NHIS-National Health Information Database; NCC, National Cancer Center; KNCI DB, Korea National Cancer Incidence database; KCDC, Korea Centers for Disease Control and Prevention; KNHANES, Korea National Health and Nutrition Examination Survey; KTBS, Korean Tuberculosis Surveillance; KoGES, Korean Genome and Epidemiology Study; NIP, National Immunization Program.
* Information based on the Healthcare Big Data platform (https://hcdl.mohw.go.kr/BD/Portal/Enterprise/DefaultPage.bzr).
Table 3.
Table 4.
| Guidelines | Publication year | Source | Checklist items | Link |
|---|---|---|---|---|
| Guide on Methodological Standards in Pharmacoepidemiology | 2018 (version 7) | ENCePP | Research question, study design, data sources, source and study population, definition and measurement of exposures/outcomes, bias, effect measure modification, data management, data analysis, quality control, ethical/data protection issues, communication of study results | http://www.encepp.eu/standards_and_guidances/methodologicalGuide.shtml |
| GPP | 2016 (version 4) | Public Policy Committee and International Society of Pharmacoepidemiology | Population, definition of exposures/outcomes/other risk factors, study size, statistical precision, data management, data analysis, quality assurance, quality control | https://doi.org/10.1002/pds.3891 |
| STROBE | 2007 (version 4) | STROBE Initiative | Introduction (background, objective), methods (study design, setting, participants, data source, bias, study size, statistical analysis), results (descriptive data, outcome, main results, other analysis), discussion (interpretation, generalizability, limitations), funding information | https://www.strobe-statement.org/index.php?id=available-checklists |
| ROBINS-I | 2016 (version 1) | Researchers, many involved with Cochrane systematic reviews | Bias related to confounding factors, selection of participants, classification of interventions, deviations from the intended interventions, missing data, measurement of outcomes, and selection of reporting results | http://dx.doi.org/10.1136/bmj.i4919 |
REFERENCES
- TOOLS







