 |
 |
- Search
| Korean J Fam Med > Volume 43(5); 2022 > Article |
|
Abstract
Background
In addition to its antidiabetic effects, metformin has pleiotropic effects, such as the inhibition of carcinogenesis. This study aimed to investigate the association between metformin use and pancreatic cancer risk in the Korean National Health Insurance Service (NHIS)-National Health Screening Cohort (HEALS).
Methods
Of the individuals in the Korean NHIS-HEALS, 29,271 men and 19,091 women were included in the final analysis after propensity score matching based on age, body mass index, and smoking status. The study population was categorized into three groups: metformin non-users with diabetes mellitus (DM), metformin users with DM, and non-diabetic users. A Cox proportional hazards regression model was used to examine the association between metformin use and pancreatic cancer.
Results
The median follow-up period was 12.9 years. The estimated pancreatic cancer incidence was highest in metformin users with DM, regardless of sex (P<0.001), and lowest in non-diabetic men and female metformin non-users (P=0.053). The hazard ratios (95% confidence interval) for pancreatic cancer incidence in metformin users and non-diabetic individuals were 1.116 (0.648–1.923) and 0.447 (0.259–0.771) in men and 2.769 (1.003–7.642) and 1.451 (0.529–3.984) in women, respectively, after full adjustment.
Metformin is a first-line treatment along with lifestyle modifications for the management of type 2 diabetes mellitus (DM), as recommended by many academic societies, including the American Diabetes Association and European Association for the Study of Diabetes [1,2]. Although the underlying mechanism of action of metformin remains unclear, its main actions seem to be the inhibition of gluconeogenesis in the liver and increasing insulin sensitivity in peripheral tissues, such as muscles [3]. According to the UK Prospective Diabetes Study, intensive treatment of early phase DM can decrease diabetes-related outcomes, such as vascular complications and death [4]. Metformin has various effects beyond glycemic control. Adenosine monophosphate (AMP)-activated protein kinase (AMPK) may mediate the pleiotropic actions of metformin, including its ability to control blood glucose levels, prevent carcinogenesis, and modulate the immune system [3,5-7]. Activated AMPK affects many downstream proteins, for example, it inhibits mammalian target of rapamycin (mTOR) and stimulates p53 [8], both of which play major roles in modulating carcinogenesis. AMPK also has an anti-inflammatory function [8]. Although the associated mechanisms remain unclear, metformin may play a role in the prevention of carcinogenesis.
Pancreatic cancer is one of the cancers with the worst prognosis. The incidence of pancreatic cancer per 100,000 people has steadily increased, from 5.6% in 1999 to 7.6% in 2021 [9]. Unfortunately, the 5-year survival rates of patients with pancreatic cancer in Korea has not improved. The 5-year relative survival rates of pancreatic cancer diagnosed in 1993–1995 and 2014–2018 were 10.6% and 12.6%, respectively [10]. The initial symptoms of pancreatic cancer only appear in the advanced stages. Even in resectable cancers, adjuvant treatments, such as chemotherapy and radiotherapy, are frequently required [11]. Currently, there is no reliable screening method for the detection of early pancreatic cancer. Therefore, prevention of pancreatic cancer is the most effective way to reduce the related mortality and burden of illness. If anti-diabetic medications, such as metformin, can play a role in preventing the development of pancreatic cancer, this would allow effective control of diabetes and a reduction in the burden of pancreatic cancer. Some previous studies have reported that metformin use was associated with a lower risk of pancreatic cancer [12,13]; however, a few studies have demonstrated a null association [14]. Because of these inconsistent findings, further research is required.
This study aimed to examine the association between metformin use and pancreatic cancer incidence by analyzing data from the Korean National Health Insurance Service (NHIS)-National Health Screening Cohort (HEALS).
Cohort data from 2002 to 2015 were obtained from the Korean Health Insurance Corporation. The cohort consisted of 514,794 random samples out of 5.15 million people between the ages of 40 and 79 years as of December 2002, who underwent medical checkups between 2002 and 2003. Data included medical information such as diagnosis code, prescription, age, sex, medical checkups, death, and monthly household income range, among other variables.
A flowchart of the exclusion conditions is shown in Figure 1. Subjects who met the following conditions were sequentially eliminated: (1) aged 70 or older as of 2003 (n=38,519), (2) received a cancer diagnosis (International Classification of Diseases, 10th revision [ICD-10] code: C00–C97 or D00–D09) between 2002 and 2004 (n=35,136), (3) a history of cancer before 2004 (n=1,218), (4) died between 2002 and 2004 (n=1,401), (5) had an initial diagnosis of diabetes between 2004 and 2015 (n=69,988), (6) were prescribed insulin for 90 days or more between 2002 and 2003 (n=63), (7) received metformin prior to diabetes diagnosis (n=1,028), (8) did not have diabetes but were prescribed metformin between 2002 and 2015 (n=2,667), (9) were prescribed metformin for 90 days or less between 2002 and 2003 but 90 days or more between 2002 and 2015 (n=21,167), (10) subjects with any incomplete values for confounding covariates (n=20,040), and (11) subjects with a study period of 30 days or less (n=137).
Finally, 323,430 subjects were included in the analysis. As there were far fewer metformin recipients than non-users, we employed propensity score matching (PSM). For PSM, the MatchIt package in R (https://cran.r-project.org/) was used. Age, body mass index (BMI), and smoking status were used as matching variables. The ratio of metformin users to metformin non-users to non-diabetic individuals was 1:1:4. The final number of participants was 29,271 males and 19,091 females.
All the procedures followed the 1964 Declaration of Helsinki. This study was approved by the Institutional Review Board of Chungbuk National University Hospital (CBNUH-2020-01-012-001). Data used in this research are available if researchers who want to deal with the Korean National Health Insurance Service (NHIS) database get permission from the Korean NHIS.
The main outcome was the occurrence of pancreatic cancer, defined as cases in which the ICD-10 code of pancreatic cancer (C25) was reported twice or more in the inpatient medical record as the main diagnosis code for hospital admission since January 2005. The date of occurrence of pancreatic cancer was considered as the day of the first record of the main diagnosis code (C25).
Subjects were defined as having diabetes if either of the following two conditions were satisfied: (1) diabetes diagnosis (ICD-10 code: E11–14) and anti-diabetic medication prescriptions, such as insulin, metformin, thiazolidinedione, dipeptidyl peptidase-4 inhibitor, α-glucosidase inhibitor, sodium-glucose cotransporter-2 inhibitor, glucagon-like peptide-1 agonist, and other anti-diabetic drugs, for more than 90 days; or (2) fasting blood glucose (FBG) level of 126 mg/ dL or higher in medical checkups. In this study, three groups were defined. The non-diabetic group was defined as individuals who did not meet the aforementioned definition of DM during the entire study period between 2002 and 2015. Metformin users comprised subjects who were diagnosed with diabetes between 2002 and 2003 and who also had prescriptions including metformin for more than 90 days between 2002 and 2003. The metformin non-users comprised subjects diagnosed with diabetes between 2002 and 2003 who took metformin for less than 90 days during the entire study period.
There was a slight difference between the groups with regard to the study period calculation. For the non-diabetic group, the start date of the study was defined as the first medical checkup date between 2002 and 2003. For both metformin users and non-users, the start date of the study was the initial occurrence of diabetes between 2002 and 2003. Generally, the end date of the study was the date of occurrence of cancer or death, whichever occurred first. If neither cancer nor death occurred, the end date of the study was the date of the most recent medical checkup or last hospital visit. For metformin users, the end date was defined as the most recent of either the last health checkup, last hospital visit, or last metformin intake date.
We considered age, BMI, systolic blood pressure (SBP), total cholesterol, FBG, alanine aminotransferase (ALT) levels, hypertension status, smoking status, drinking status, physical activity, and income level as possible confounding factors. Among these, the hypertension status, smoking status, drinking status, physical activity, and income level were converted into categorical variables. Hypertension was divided into two categories: presence or absence of a history of hypertension. Subjects were defined as non-smokers (individuals who had never smoked before) or ever smokers (individuals who had currently quit cigarette smoking or still smoked cigarettes during the relevant survey period). Drinking status was classified as rare (individuals who drank alcohol less than 2 days a month), sometimes (individuals who drank alcohol 2 days a month to 2 days a week), or regular (individuals who drank alcohol 3 or more days a week). Physical activity was categorized as rare (individuals who engaged in exercise less than once a week), sometimes (individuals who engaged in exercise from 1–4 days a week), or regular (individuals who engaged in at least 5 days of exercise per week). The monthly household income level was categorized as low (0–30th percentile), middle (31–70th percentile), or high (71– 10th percentile).
For group comparisons, we used analysis of variance for continuous variables and chi-square analysis for categorical variables. The KaplanMeier method and log-rank test were used to estimate and compare the survival rates of the three groups. A Cox proportional hazard regression model (Cox PH model) was used to determine the hazard ratios (HRs) of the three groups. Four adjustment models were considered for the Cox PH model: (1) age in model 1; (2) age, smoking status, drinking status, and physical activity in model 2; (3) BMI, SBP, total cholesterol, ALT, hypertension status, and monthly household income level, in addition to the variables of model 2 in model 3; and (4) FBG level in addition to the variables of model 3 in model 4. SAS enterprise guide ver. 7.1 (SAS Institute Inc., Cary, NC, USA) and R studio ver. 3.3.3 (RStudio, Boston, MA, USA) were used for statistical analyses. A two-tailed P-value of <0.05 was considered statistically significant.
During the study period (median follow-up of 12.9 years), 182 cases of pancreatic cancer (130 men and 52 women) were diagnosed in 48,362 individuals (23,271 men and 19,091 women), with a cumulative incidence of 0.37% in the entire population (0.44% in men and 0.27% in women).
The baseline characteristics of the participants according to the presence or absence of diabetes and metformin use after PSM using age, BMI, and smoking status are shown in Table 1. The mean age was not different between males and females in the three groups. Even with BMI matching, male metformin users were mostly obese (P= 0.027), whereas there was no difference among the female groups. Blood glucose and ALT levels were highest in metformin users of both sexes, while SBP and total cholesterol levels were higher in metformin non-users (all P<0.05). The percentage of individuals who consumed alcohol more frequently was lowest among both male and female metformin users (P<0.05).
The relationship between metformin use and pancreatic cancer incidence is presented in Figure 2 using Kaplan-Meier survival curves. The P-values from the log-rank test were P<0.001 for men and P=0.052 for women. The difference in cumulative incidence by group was significant in men but only marginally significant in women. In the male DM population, metformin users had a cumulative incidence as high as that of metformin non-users (P=0.735). The male non-diabetic group had a lower cumulative incidence than male metformin users and non-users (P<0.05). In the female DM population, the cumulative incidence of metformin use was much higher than that of metformin non-users (P=0.019). Metformin non-users had a similar cumulative incidence as the non-diabetic group (P=0.397).
Table 2 shows the Cox PH regression models and HRs (95% confidence intervals [CIs]) for the incidence of pancreatic cancer. Compared to metformin non-users, the HR (95% CI) for pancreatic cancer incidence in metformin users was 1.103 (0.645–1.887) in men and 2.894 (1.058–7.916) in women after adjusting for age, while that in nondiabetic individuals was 0.436 (0.266–0.714) in men and 1.585 (0.619– 4.060) in women (model 1). After adjusting for age, health behaviors (cigarette smoking, alcohol consumption, and physical activity), BMI, SBP, total cholesterol, ALT levels, hypertension history, and monthly household income, the HR (95% CI) for pancreatic cancer incidence in metformin users with DM and non-diabetic individuals were 1.118 (0.650–1.923) and 0.444 (0.269–0.732) in men and 2.772 (1.004–7.648) and 1.511 (0.587–3.891), respectively, in women (model 3). To minimize hyperglycemic effects, blood glucose levels were adjusted in model 4. The HRs (95% CIs) for pancreatic cancer incidence in metformin users and non-diabetic individuals were 1.116 (0.648–1.923) and 0.447 (0.259–0.771) in men, and 2.769 (1.003–7.642) and 1.451 (0.529– 3.984) in women, respectively.
In the male diabetic population, the incidence rates of pancreatic cancer were similar in metformin users and non-users based on the analysis of the NHIS-HEALS data. However, non-diabetic men had a lower risk of pancreatic cancer than metformin non-users. In contrast, metformin use increased the risk of pancreatic cancer in the female diabetic population.
Several studies have demonstrated that metformin reduces the risk of various cancers [15,16]. However, there are conflicting results regarding the association between metformin use and the incidence of pancreatic cancer. Tseng et al. [12] reported that metformin ever-users were at a lower risk of pancreatic cancer after PSM. Liao et al. [14] revealed a null association between metformin and pancreatic cancer based on a population-based analysis of the Taiwan National Health Insurance Database. A Korean study supported the possibility that metformin can reduce the risk of pancreatic cancer after PSM [13]. Based on a systematic review and meta-analysis, Wang et al. [17] reported that metformin use is associated with a lower risk of pancreatic cancer. In addition to cancer prevention, metformin has survival benefits in patients with pancreatic cancer [18]. In animal studies, metformin has been shown to suppress the initiation, progression, and invasiveness of pancreatic cancer [19]. In contrast to these studies, Ferrara et al. [20] demonstrated that metformin users were at a higher risk of pancreatic cancer than individuals who used pioglitazone. Furthermore, Lu et al. [21] reported that metformin use increases pancreatic cancer risk.
Our results indicated that metformin use in female patients with diabetes can increase the risk of pancreatic cancer. The exact mechanism by which metformin increases the risk of pancreatic cancer is unknown. Previous studies have reported that metformin can inhibit the development of pancreatic cancer. Metformin changes the interaction between the insulin receptor and other growth factor signaling systems in human pancreatic cancer cells [22]. Metformin inhibits the growth of human pancreatic cancer cells by inhibiting the mTOR activity [23]. These mechanisms support the finding that metformin reduces the risk of pancreatic cancer. However, our results were in direct opposition to this. We still do not understand the exact mechanism by which metformin increases the risk of pancreatic cancer; this may simply be an epiphenomenon. As an example of this mechanism, the positive relationship between metformin use and pancreatic cancer risk could be caused by the contamination of metformin products. The U.S. Food & Drug Administration has been investigating the presence of nitrosamines such as N-nitrosodimenthylamine (NDMA) in some metformin products [24,25]. Long-term exposure to carcinogenic NDMA levels above the safety range may increase the risk of cancer.
Our findings are inconsistent with our hypothesis that metformin can reduce pancreatic cancer. In addition, these findings contradict those of Lee et al. [13], although both studies were based on the Korean population. In part, opposing outcomes might be caused due to the use of different Korean insurance claims data; our study contained health screening information such as laboratory findings, whereas the data used by Lee et al. [13] did not. The median follow-up duration of our study was longer than that in a study by Lee et al. [13] (12.9 years versus 4.9 years). The relatively longer median follow-up period in our study may have resulted in more accurate results than a short follow-up period, because carcinogenesis has a long-time course from exposure to diagnosis. In our study, we matched the three groups by propensity scores based on age, BMI, and smoking status. The population was stratified by sex and further adjusted for confounding factors, such as BMI, ALT, cholesterol, and glucose levels. This strict control of parameters likely improved our model fit, and therefore, the ability to investigate the association between metformin use and pancreatic cancer. The pancreatic cancer incidence was more conservatively defined as two or more entries in the medical records of the main diagnosis code for pancreatic cancer during hospitalization. Thus, we were unlikely to have included any false-positive diagnoses to rule out pancreatic cancer in our study. As expected, we found that non-diabetic men were at a lower risk of pancreatic cancer than male metformin non-users with DM, which supports the robustness of our data.
This study has several strengths. The NHIS-HEALS cohort comprised real-world measurements from a healthcare setting. Sociodemographic information, such as health behaviors, monthly household income, and past medical histories, were included in this data. This information is closely associated with accessibility to health care and health outcomes. In addition, laboratory results, such as blood glucose levels, lipid profiles, and liver enzyme levels, and screening results for the five most common cancers, were available for this cohort. Thus, we were able to adjust for possible confounding factors and use conservative exclusion criteria, such as a history of previous cancer.
Several limitations of this study should be considered when interpreting its results. Not all factors for pancreatic cancer could be completely controlled because the relevant information was not included in the NHIS-HEALS database. Well-known risk factors for pancreatic cancer include cigarette smoking, exposure to toxic materials (chemicals and heavy metals), heavy alcohol consumption, obesity, high red/ processed meat consumption, a medical history of chronic pancreatitis, and abnormal glucose metabolism [26]. Genetic predispositions are also important. Pancreatic cancer is thought to have a familial component in approximately 10% of all cases [27]. Although we adjusted for several risk factors, including BMI, smoking status, alcohol consumption, physical activity, and FBG levels, other risk factors, including unhealthy diet (e.g., high red/processed meat consumption), exposure to toxins, and medical history of chronic pancreatitis, were not completely controlled because this information was not available from the secondary data that the NHIS collected and provided. Although pancreatic cancer was conservatively defined, misclassification was possible. Different histotypes of pancreatic cancer were not considered, because this information was not recorded in the NHIS-HEALS database. In addition, we could not control for the duration of diabetes or the level of glycated hemoglobin. The cumulative effects of metformin on the development of pancreatic cancer were not assessed because of the small population size of patients with pancreatic cancer. Determining cumulative effects could have resulted in a more accurate identification of causal relationships. Drug combination therapy for intensive glycemic control is becoming increasingly important in the treatment of diabetes [4]. Therefore, it is difficult to investigate the association between specific antidiabetic drugs and pancreatic cancer risk. Additional studies are needed on the interaction between metformin and other antidiabetic agents, which could affect the development of pancreatic cancer. Finally, we did not investigate the mechanism by which metformin increased the risk of pancreatic cancer. Therefore, further research is required in this regard.
In summary, men without DM had a lower risk of pancreatic cancer than male metformin non-users with DM, whereas female metformin users with DM were at a higher risk of pancreatic cancer than female metformin non-users with DM.
Figure. 1.
Flowchart of the inclusion and exclusion criteria. NHIS-HEALS, National Health Insurance Service-National Health Screening Cohort; PSM, propensity score matching; DM, diabetes mellitus.
Figure. 2.
(A, B) Estimated cumulative incidence of pancreatic cancer P-values from the log-rank tests. Each number means the cumulative incidence rate as percentage. Cumulative incidence is 1-survival rate. P-value is from the log-rank test.
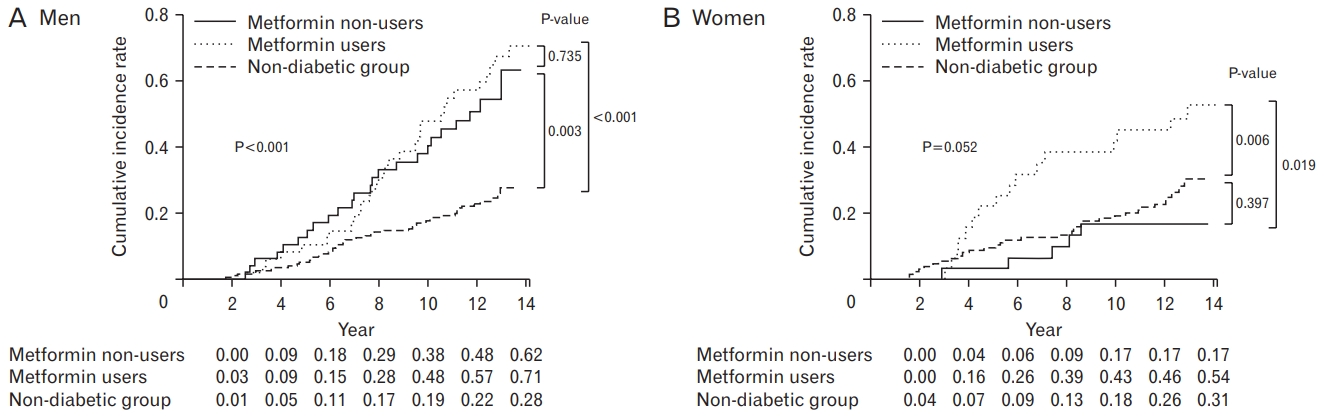
Table 1.
Baseline characteristics of the study population after propensity score matching
Table 2.
Cox-proportional hazard regression models for pancreatic cancer incidence
Values are presented as hazard ratio (95% confidence interval). Model 1: adjusted for age; model 2: adjusted for smoking status, alcohol consumption, and physical activity in addition to variable in model 1; model 3: adjusted for body mass index, systolic blood pressure, serum total cholesterol level, serum alanine aminotransferase level, past hypertension history, and monthly household income, in addition to variables in model 2; and model 4: adjusted for blood glucose levels, in addition to variables in model 3.
REFERENCES
1. American Diabetes Association. 9. Pharmacologic approaches to glycemic treatment: standards of medical care in diabetes-2019. Diabetes Care 2019;42(Suppl 1):S90-102.



2. Davies MJ, D’Alessio DA, Fradkin J, Kernan WN, Mathieu C, Mingrone G, et al. Management of hyperglycaemia in type 2 diabetes, 2018: a consensus report by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetologia 2018;61:2461-98.



3. Rena G, Hardie DG, Pearson ER. The mechanisms of action of metformin. Diabetologia 2017;60:1577-85.




4. UK Prospective Diabetes Study (UKPDS) Group. Effect of intensive blood-glucose control with metformin on complications in overweight patients with type 2 diabetes (UKPDS 34). Lancet 1998;352:854-65.


5. Madiraju AK, Erion DM, Rahimi Y, Zhang XM, Braddock DT, Albright RA, et al. Metformin suppresses gluconeogenesis by inhibiting mitochondrial glycerophosphate dehydrogenase. Nature 2014;510:542-6.




6. McIntyre HD, Ma A, Bird DM, Paterson CA, Ravenscroft PJ, Cameron DP. Metformin increases insulin sensitivity and basal glucose clearance in type 2 (non-insulin dependent) diabetes mellitus. Aust N Z J Med 1991;21:714-9.


7. Viollet B, Guigas B, Sanz Garcia N, Leclerc J, Foretz M, Andreelli F. Cellular and molecular mechanisms of metformin: an overview. Clin Sci (Lond) 2012;122:253-70.




9. Jung KW, Won YJ, Hong S, Kong HJ, Im JS, Seo HG. Prediction of cancer incidence and mortality in Korea, 2021. Cancer Res Treat 2021;53:316-22.




10. Ministry of Health and Welfare; Korea Central Cancer Registry; National Cancer Center. Annual report of cancer statistics in Korea in 2018. Sejong: Ministry of Health and Welfare; 2018.
11. Tempero MA, Malafa MP, Al-Hawary M, Asbun H, Bain A, Behrman SW, et al. Pancreatic adenocarcinoma, version 2: 2017, NCCN clinical practice guidelines in oncology. J Natl Compr Canc Netw 2017;15:1028-61.


12. Tseng CH. Metformin and pancreatic cancer risk in patients with type 2 diabetes. Pancreas 2018;47:e57-9.



13. Lee DY, Yu JH, Park S, Han K, Kim NH, Yoo HJ, et al. The influence of diabetes and antidiabetic medications on the risk of pancreatic cancer: a nationwide population-based study in Korea. Sci Rep 2018;8:9719.




14. Liao KF, Lai SW, Li CI, Chen WC. Diabetes mellitus correlates with increased risk of pancreatic cancer: a population-based cohort study in Taiwan. J Gastroenterol Hepatol 2012;27:709-13.


15. Kim YI, Kim SY, Cho SJ, Park JH, Choi IJ, Lee YJ, et al. Long-term metformin use reduces gastric cancer risk in type 2 diabetics without insulin treatment: a nationwide cohort study. Aliment Pharmacol Ther 2014;39:854-63.


16. Ruiter R, Visser LE, van Herk-Sukel MP, Coebergh JW, Haak HR, Geelhoed-Duijvestijn PH, et al. Lower risk of cancer in patients on metformin in comparison with those on sulfonylurea derivatives: results from a large population-based follow-up study. Diabetes Care 2012;35:119-24.

17. Wang Z, Lai ST, Xie L, Zhao JD, Ma NY, Zhu J, et al. Metformin is associated with reduced risk of pancreatic cancer in patients with type 2 diabetes mellitus: a systematic review and meta-analysis. Diabetes Res Clin Pract 2014;106:19-26.


18. Wirunsawanya K, Jaruvongvanich V, Upala S. Survival benefits from metformin use in pancreatic cancer: a systemic review and meta-analysis. Pancreas 2018;47:e11-4.


19. Chen K, Qian W, Jiang Z, Cheng L, Li J, Sun L, et al. Metformin suppresses cancer initiation and progression in genetic mouse models of pancreatic cancer. Mol Cancer 2017;16:131.




20. Ferrara A, Lewis JD, Quesenberry CP Jr, Peng T, Strom BL, Van Den Eeden SK, et al. Cohort study of pioglitazone and cancer incidence in patients with diabetes. Diabetes Care 2011;34:923-9.




21. Lu Y, Garcia Rodriguez LA, Malgerud L, Gonzalez-Perez A, MartinPerez M, Lagergren J, et al. New-onset type 2 diabetes, elevated HbA1c, anti-diabetic medications, and risk of pancreatic cancer. Br J Cancer 2015;113:1607-14.




22. Kisfalvi K, Eibl G, Sinnett-Smith J, Rozengurt E. Metformin disrupts crosstalk between G protein-coupled receptor and insulin receptor signaling systems and inhibits pancreatic cancer growth. Cancer Res 2009;69:6539-45.




23. Rozengurt E. Mechanistic target of rapamycin (mTOR): a point of convergence in the action of insulin/IGF-1 and G protein-coupled receptor agonists in pancreatic cancer cells. Front Physiol 2014;5:357.



24. Parr MK, Joseph JF. NDMA impurity in valsartan and other pharmaceutical products: analytical methods for the determination of N-nitrosamines. J Pharm Biomed Anal 2019;164:536-49.


25. U.S. Food and Drug Administration. Statement from Janet Woodcock, M.D., director of FDA’s Center for Drug Evaluation and Research, on impurities found in diabetes drugs outside the U.S. [Internet]. Silver Spring (MD): U.S. Food and Drug Administration; 2019 [cited 2021 Apr 24]. Available from: https://www.fda.gov/news-events/press-announcements/statement-janet-woodcock-md-director-fdas-center-drug-evaluation-and-research-impurities-found







