|
 |
- Search
| Korean J Fam Med > Volume 43(2); 2022 > Article |
|
Abstract
Systemic lupus erythematosus (SLE) is a connective tissue disease of unknown etiology that predominantly affects women of childbearing age. We report a case of male systemic lupus erythematous with antinuclear antibodies and typical clinical presentations of multiple skin lesions, polyarticular joint pain, fatigue, anorexia, and hair loss. Full evaluations were used to establish a diagnosis of SLE. The lower prevalence of SLE among males and antinuclear antibody-negative patients poses a great challenge for diagnosis. Therefore, as primary care doctors, we need to have a high suspicion of systemic lupus erythematous even in male and antinuclear antibody-negative patients. Thus, early treatment may help patients improve their quality of life.
Systemic lupus erythematosus (SLE) is a connective tissue disease of unknown etiology, and it is generally common among women of childbearing age and rare among men [1]. The reported female to male ratio is 8–15:1; pre-pubertal and post-menopausal ratios are much lower at 2–6:1 and 3–8:1, respectively [2]. The prevalence of SLE in Malaysia was approximately 43/100,000 populations. The prevalence of ethnic Chinese, Malays, and Indians were 46, 26, and 12 per 100,000 populations, respectively, with an overall female to male ratio of 12:1 [3].
Hormonal factors are associated with disease pathogenesis, but a full explanation for the rarity of the disease in men remains elusive [4]. Symptoms, including skin rash, extreme fatigue, and joint pain, are similar in men and women, but men have a more complex clinical course [1]. The diagnosis of SLE is based on both clinical and immunologic criteria, and antinuclear antibodies (ANA) (90.9%) are the most common serological markers identified. Dey et al. [4] reported that 91.7% of male SLE cases in Ghana were ANA-positive, 33.3% were anti-double-stranded DNA (anti-dsDNA)-positive, 58.3% were extractable nuclear antigens, and 58.3% had ribosomal antibodies.
We report a case of an ANA-negative 22-year-old man who improved clinically after undergoing treatment. Although SLE is less common in men and ANA-negative patients, it should still be considered as a differential diagnosis. Early recognition and intervention appear to be associated with decreased morbidity.
A 22-year-old male contractor worker presented with a three-month history of multiple skin lesions that started over the bilateral arms and spread to the face, neck, and lower limbs. The skin lesions were painless and non-itchy. They started as erythematous plaques and became hyperpigmented within a week. He also had multiple joint pain, lethargy, loss of appetite, and loss of weight. The joint pain involved the bilateral metacarpal phalangeal, proximal interphalangeal, elbow, shoulder, knee, and ankle. It was associated with hand stiffness that lasted for 1 hour in the morning. One month later, the patient presented with hair loss. Otherwise, he had no family history of autoimmune disease. He denied any high-risk behavior or tuberculosis exposure.
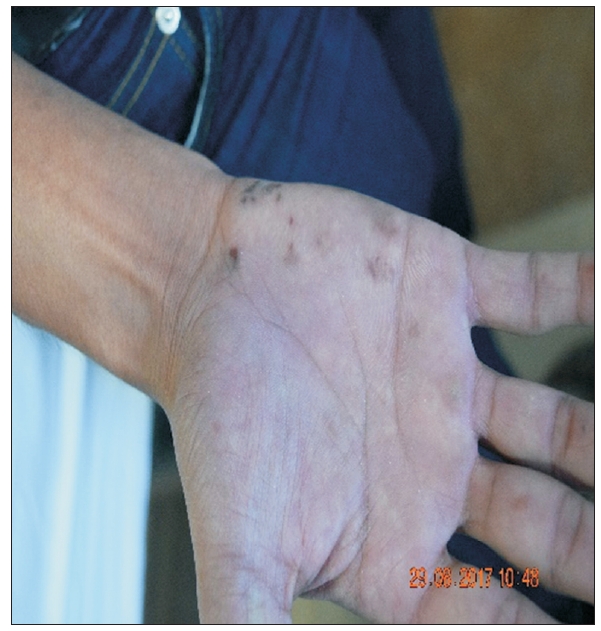
Physical examination showed that the patient was hemodynamically stable. The skin showed hyperpigmented plaques over his malar prominence, sparing the nasolabial fold and adherent keratotic scales, followed by atrophic scarring involving the chest, back, arms, and legs (Figure 1). Vasculitis spots were observed over the palms and soles (Figure 2), and non-scarring alopecia was observed over the temporal scalp (Figure 3). Joint examinations revealed mild tenderness over the metacarpophalangeal joints and elbow joints with no swelling, redness, or limitation of joint movement.
During the first visit to a health clinic, a work-up was performed for possible SLE. Other differential diagnoses include photo-aggravated seborrheic dermatitis, contact dermatitis, dermatomyositis, rheumatoid arthritis, and lupus vulgaris. He was prescribed topical steroids for the skin lesions and analgesia (non-steroidal anti-inflammatory drug) for joint pain. After 2 weeks, the patient was followed up and did not show much improvement. Initial blood tests revealed no significant findings. Complete blood count revealed white blood cell 8.16×109/L, hemoglobin 156 g/L, and platelet 238×109/L. Urinalysis was normal. ANA was negative; hence, anti-dsDNA antibody testing was not conducted. Rheumatoid factor was negative. Renal function was normal. C3 was low at 0.62 g/L (normal range, 0.66–1.3 g/L) and C4 was 0.23 g/L (normal range, 0.2–0.6 g/L). The erythrocyte sedimentation rate was 19, and human immunodeficiency virus enzyme-linked immunosorbent assay was non-reactive. The case was discussed with the medical team; however, due to incomplete criteria, SLE could not be confirmed.
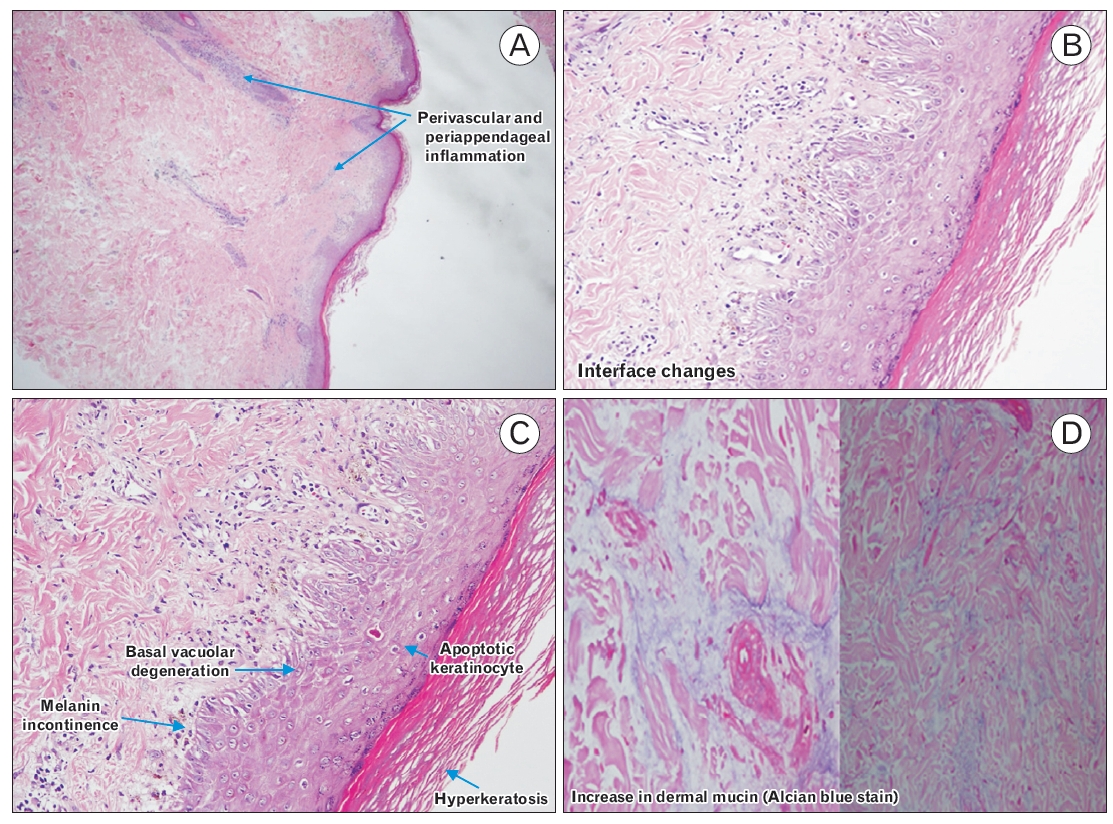
The patient was followed up at a health clinic. At this time, he presented with hair loss, persistent skin lesions, and joint pain. The patient was arranged for a skin biopsy for further investigation after discussion with the medical team. Skin biopsy was performed by the dermatology team (Figure 4), and immunofluorescence revealed dermoepidermal deposition of immunoglobulin G, immunoglobulin M, C3c, and Clq, which are features of lupus erythematosus. An anti-extractable nuclear antigen screening test showed the presence of antibodies to the Sm nuclear antigen, as well as SSA/Ro and RNP. Jo-1 and Scl 70 were negative. He fulfilled five of 17 of the Systemic Lupus International Collaborating Clinics (SLICC) criteria for the diagnosis of SLE, which included clinical criteria for acute cutaneous lupus, non-scarring alopecia, and joint disease and immunologic criteria of positive anti-Sm and low C3.
The patient was started on prednisolone 25 mg tablets once a day (weight 50 kg, 0.5 mg/kg) during the first encounter with a dermatologist. He was, subsequently, started on oral hydroxychloroquine 200 mg daily after the diagnosis of SLE. Oral prednisolone was tapered to 5 mg daily. The patient was also prescribed sunblock SPF 50 daily over the limbs and face, betamethasone valerate cream 1:4 daily over the skin lesions, and betamethasone valerate ointment over the scalp area. A referral for eye assessment was made. Subsequent follow-up showed that the patient had marked improvement in the skin lesions, and he had no more joint pain.
The patient provided written informed consent for the publication of details and images.
SLE is a clinically heterogeneous autoimmune disease of unknown etiology. It is characterized serologically by autoantibodies that target self-proteins. Organs and cells of affected individuals undergo damage mediated by tissue-binding autoantibodies and immune complexes [4]. Although the precise etiology of SLE remains vague, genetic predisposition and environmental and hormonal factors are deemed to play important roles in its pathogenesis [2]. Females are more commonly affected by SLE, and an estrogen effect has been indicated by several observations for this difference [5]. Women exposed to estrogen-containing oral contraceptives or hormone replacement have an increased risk of developing SLE. Females of several mammalian species are known to demonstrate higher antibody responses than males [5]. Hughes et al. [6] reported sex-specific genetic differences in SLE, in which significant gene-sex interactions were observed in the human leukocyte antigen gene region and IRF5 gene. A genetic effect unique to women was also observed in KIAA1542. The study demonstrated that men with SLE have a higher cumulative genetic risk than women, and men need to inherit a larger number of risk alleles to develop SLE than women. These findings suggest that sex disparities in SLE can be at least partly related to factors beyond the X chromosome and hormonal differences [6].
SLE diagnosis is based on clinical and laboratory findings. However, even today, there is often a significant delay between the onset of symptoms and diagnosis. The average duration from symptom onset to the diagnosis of SLE is approximately 2 years, despite increasing awareness of SLE. This is probably due to the lower suspicion and a longer time lag reported for children, males, and late-onset disease [7]. Similarly, we had diagnostic difficulties during the initial presentation. He had multiple visits to the primary care center for his symptoms before the diagnosis of SLE was made. Patients with SLE may present with various clinical manifestations and lupus mimickers. Lupus mimickers refer to a group of conditions that exhibit clinical and laboratory features resembling SLE and include infections, neoplasms, and medications. Several overlapping syndromes combine SLE characteristics with features from other diseases, including rheumatoid arthritis (also referred to as “rhupus”), polymyositis/dermatomyositis, systemic sclerosis, and Sjögren’s syndrome [7].
Symptoms of SLE are similar in men and women, particularly skin rash, extreme fatigue, and joint pain. However, the findings suggest that the disease in men has a more complex clinical course, and some studies have shown that renal impairment, central nervous system, and vascular disease are more common in men than in women [1]. Male SLE patients in Ghana are comparable to other populations, with arthritis and constitutional features being predominant early features and lupus nephritis being the main early indicator of organ damage [4]. Murphy and Isenberg [2] reported consistent evidence for reduced incidence of alopecia, malar rash, and arthralgia/arthritis in men at presentation and during the subsequent disease course. In contrast, it cannot be reliably said that there is a definite increase in the incidence of nephropathy, thrombotic episodes, and, most importantly, a greater mortality risk [2]. Our patient experienced cutaneous lesions, arthralgia, and alopecia, which are commonly present at diagnosis in women. Although our patient presented with typical clinical presentations of SLE, the physicians faced a diagnostic dilemma due to the gender predilection and ANA-negativity.
The diagnosis of SLE in our patient was made based on the 2012 SLICC criteria. He had to satisfy at least four of the 17 criteria, including at least one of the 11 clinical criteria and one of the six immunologic criteria, or have biopsy-proven nephritis compatible with SLE in the presence of ANA or anti-dsDNA antibodies. Our patient fulfilled five of 17 ANA-negative criteria. In a Scandinavian study of confirmed SLE cases and individuals with lupus-mimicking conditions, the SLICC classification showed comparable diagnostic sensitivity, specificity, and accuracy to the latest classification of European League Against Rheumatism/American College of Rheumatology [8]. Of a panel of international lupus experts, 27% felt comfortable diagnosing SLE even in the absence of positive ANA [7]. ANAs have a sensitivity of 33.6%, while anti-dsDNA has a sensitivity of 57.1%, indicating that a significant portion of SLE cases may be missed based on these markers alone. Studies have suggested that a positive ANA may disappear in some SLE patients over time, with sensitivity dropping to 76% and positivity dropping from 98% to 71% in patients with established SLE [9]. Khajehdehi et al. [10] reported a rare case of class III lupus nephritis in a male who developed clinical flare-up of SLE despite being ANA-negative. Zhao [9] suggested that more biomarkers may be associated with SLE, given the strongly positive nonspecific anti-neutrophil antibody in the setting of marker negativity.
Glucocorticoids and antimalarial drugs are the mainstay of lupus management, along with immunosuppressive or biologic drugs. Emerging evidence suggests that dosage should not exceed 5 mg/d of prednisone for chronic maintenance therapy for SLE. Current recommendations also suggest that antimalarial drugs, such as hydroxychloroquine, should be considered in all SLE patients as it not only controls disease activity but also reduces damage accrual and mortality rates [7]. Our patient improved markedly after starting oral hydroxychloroquine 200 mg daily and tapering the dose of prednisolone.
This case report aims to increase awareness among primary care doctors, who should always consider SLE as a diagnosis in patients presenting with systemic symptoms regardless of gender and the possibility of ANA-negativity. The shared care and collaboration between different disciplines and levels of care are also important to overcome uncertainties and difficulties in the clinical diagnosis and management of SLE. This is crucial because it leads to early recognition, diagnosis, and treatment, which, in turn, reduces morbidity and increases the quality of life of patients.
ACKNOWLEDGMENTS
We would like to thank Department of Dermatology and Department of Pathology of Hospital Raja Perempuan Zainab II, Kelantan, Malaysia for the support.
Figure. 1.
Hyperpigmented atrophic lesions with scarring of the malar distribution over the cheek and nose (butterfly rash) (A) and arm (B). Written informed consent for publication of this image was obtained from the patient.

Figure. 2.
Vasculitis spots over palms. Written informed consent for publication of this image was obtained from the patient.
REFERENCES
1. do Socorro Teixeira Moreira Almeida M, da Costa Arcoverde J, Barros Jacobino MN, Coimbra Neto AR. Male systemic lupus erythematosus, an overlooked diagnosis. Clin Pract 2011;1:e103.



2. Murphy G, Isenberg D. Effect of gender on clinical presentation in systemic lupus erythematosus. Rheumatology (Oxford) 2013;52:2108-15.


3. Wang F, Wang CL, Tan CT, Manivasagar M. Systemic lupus erythematosus in Malaysia: a study of 539 patients and comparison of prevalence and disease expression in different racial and gender groups. Lupus 1997;6:248-53.


4. Dey D, Ofori E, Hutton-Mensah KA, Akutek M, Okine R, Amoaba I, et al. Clinical characteristics of males with systemic lupus erythematosus (SLE) in an inception cohort of patients in Ghana. Ghana Med J 2019;53:2-7.



5. Agrawaal KK, Dhakal SS. Systemic lupus erythematosus in males: a case series. Saudi J Kidney Dis Transpl 2014;25:638-42.


6. Hughes T, Adler A, Merrill JT, Kelly JA, Kaufman KM, Williams A, et al. Analysis of autosomal genes reveals gene-sex interactions and higher total genetic risk in men with systemic lupus erythematosus. Ann Rheum Dis 2012;71:694-9.



7. Gergianaki I, Bertsias G. Systemic lupus erythematosus in primary care: an update and practical messages for the general practitioner. Front Med (Lausanne) 2018;5:161.



8. Dahlstrom O, Sjowall C. The diagnostic accuracies of the 2012 SLICC criteria and the proposed EULAR/ACR criteria for systemic lupus erythematosus classification are comparable. Lupus 2019;28:778-82.



- TOOLS