 |
 |
- Search
| Korean J Fam Med > Volume 43(4); 2022 > Article |
|
Abstract
Background
The association between life-event stressors and low physical activity linked to the severity of coronary artery disease (CAD) is poorly understood. Therefore, this study aimed to determine the perceived stress status and physical activity level associated with the severity of CAD.
Methods
The study was conducted among 423 patients with newly discovered CAD (both sexes, aged 35ŌĆō65 years) confirmed by coronary angiography results. CAD severity was classified according to the Gensini score as severe or non-severe. The fasting blood glucose and lipid profiles were also investigated. Anthropometric and brachial blood pressure measurements were obtained. A structured questionnaire including participantsŌĆÖ characteristics, the Perceived Stress Scale (PSS-14), and the International Physical Activity Questionnaire were used via face-to-face interviews. Multivariable binary logistic regression was used to assess the predictors of CAD severity using IBM SPSS ver. 24.0 (IBM Corp., Armonk, NY, USA).
Results
Most participants were classified as having severe CAD (63.8%). Adjusting for age, sex, smoking status, systolic blood pressure, body mass index, and history of hypertension, the severity of CAD increased by 9% for a onescore increase in the PSS (odds ratio [OR], 1.09; 95% confidence interval [CI], 1.01ŌĆō1.14; P=0.030). Moreover, sufficiently active (600ŌĆō1,500 metabolic equivalents [METs]/wk) patients had lower odds of severe CAD (OR, 0.43; 95% CI, 0.23ŌĆō0.72; P=0.027) than those with insufficient activity. However, no significant association was found between physical activity (Ōēź1,500 METs/wk) and CAD severity.
The association between coronary artery disease (CAD) and conventional risk factors including mental health problems and physical activity (PA) is universally well-recognized [1]. However, the association between these risk factors and CAD severity is less consistent. CAD is one of the leading causes of mortality and morbidity among cardiovascular diseases (CVDs). Among all deaths worldwide, 31% died from CVDs in 2016 [2]. In Palestine, the prevalence of CAD in 2019 was the highest (8.3%) among the CVDs in the Gaza Strip, followed by that of stroke events (3%) [3].
According to Marie et al. [4], about one-third of Palestinians need mental health interventions. Anxiety is a serious mental health problem that is prevalent in approximately 19% of CAD patients in Palestine [5]. Stressors and the fear of the unknown are the main causes of anxiety. The magnitude of stress can be measured using different methods. The Perceived Stress Scale (PSS) is a popular stress measurement tool. The concept of PSS is that individuals self-report their level of stress in reaction to potentially threatening events [6]. In addition, psychological disturbances have been increasingly associated with the prognosis of CAD [1].
The suggested underlying mechanisms of coronary atherosclerosis are linked to cumulative psychological stressors that increase sympathetic tone, resulting in activation of adrenal cortisol, release of inflammatory mediators, induction of platelet activation, and acceleration of atherogenesis plugs. Another mechanism is related to adrenal catecholamines; they are linked to increased cyclic adenosine monophosphate production and trigger the release of free radicals, which is an initial step in the formation of the atherosclerotic plug [7].
The link between PA and CAD was first established in the early 1950s, and since then, population studies have consistently found high levels of PA to be associated with a reduced risk of CAD morbidity and mortality. The mechanisms of the effect of PA on CAD risk are not fully understood, but it is possible that they involve changes in lipid and lipoprotein metabolism [8].
Traditional risk factors including age, sex, hypertension, dyslipidemia, diabetes mellitus (DM), smoking habits, family history of CAD, and obesity are known to be associated with CAD and its severity [3]. However, the relationship between the severity of CAD and perceived stress and PA levels has not yet been confirmed. Therefore, this study aimed to investigate the association between perceived stress and PA levels and the severity of CAD among patients with newly discovered CAD.
This cross-sectional study was conducted among 423 newly diagnosed CAD patients recruited from Al-Shia Hospital, the main hospital in Gaza City, between May 2018 and July 2019.
The study protocol was approved by the Ethics Committee of Tehran University of Medical Sciences (Code: IR.TUMS.VCR.REC.1397. 636) and the Palestinian Health Research Council (Helsinki Ethical Committee of Research Code: PHRC/HC/335/18). Written informed consent was obtained from all the participants.
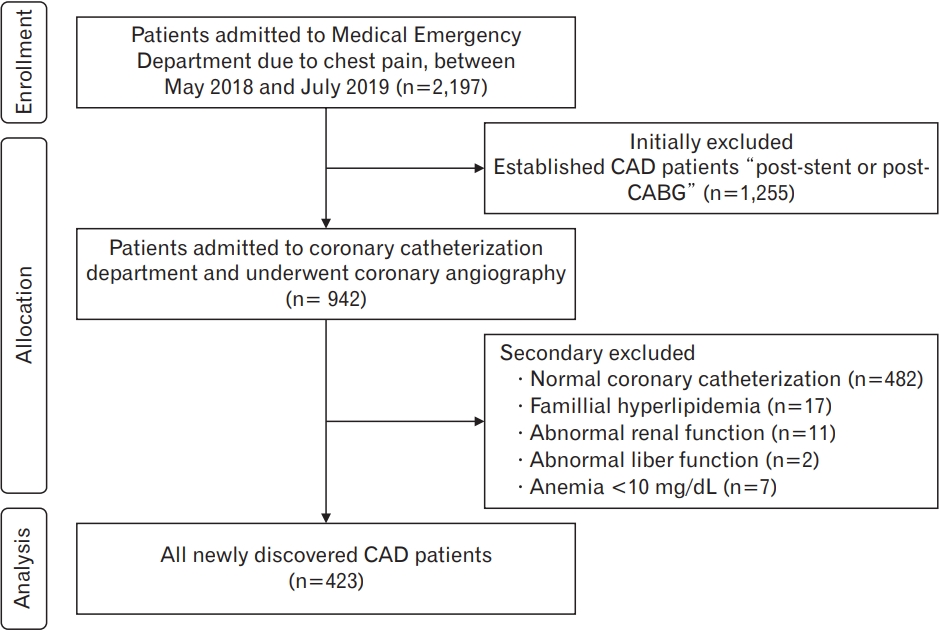
All patients with newly discovered CAD aged between 35 and 65 years who presented with chest pain and underwent coronary angiography were eligible for this study (Figure 1). The diagnosis of CAD was confirmed by two interventional cardiologists. However, patients with normal coronary angiography findings and established CAD (post-stent or post-coronary artery bypass graft [CABG]), renal disease, hepatic dysfunction, anemia or blood disorders, and familial hyperlipidemia were excluded from the study.
Based on baseline diagnostic coronary angiography, the severity of CAD was assessed using the Gensini score [9]. The Gensini score was calculated in three steps. First, a severity score was calculated for each coronary branch occlusion as follows: 1 point for Ōēż25% occlusion, 2 points for 26%ŌĆō50% occlusion, 4 points for 51%ŌĆō75% occlusion, 8 points for 76%ŌĆō90% occlusion, 16 points for 91%ŌĆō99% occlusion, and 32 points for total occlusion. For the next step, each occlusion score was multiplied by a factor according to the coronary branch (5 for the left main coronary branch; 2.5 for the proximal segment of the left anterior descending coronary branch; 2.5 for the proximal circumflex branch; 1.5 for the mid of the left anterior descending coronary branch; 1.0 for the right coronary artery, distal of the left anterior descending coronary branch, posterolateral branch, and obtuse marginal branch, and 0.5 for other branches). Finally, the Gensini score was calculated by adding the individual coronary scores. The cases were divided into two groups: severe (Gensini score Ōēź20) and non-severe CAD (Gensini score <20) [9].
An interview-based questionnaire was used, including patient characteristics, medical history, the Perceived Stress Scale (PSS-14), the International Physical Activity Questionnaire (IPAQ), anthropometric and blood pressure (BP) measurements, biochemical analysis, and the Gensini score for evaluating CAD severity. All measurements were standardized and validated. The data were collected by qualified data collectors who were provided explanations and trained to collect the data while respecting confidentiality.
Body weight was measured using a digital scale (Seca 808; Seca, Hamburg, Germany) to the nearest of 0.1 kg, with light clothes and no shoes. Height was measured using a stadiometer (patients barefooted and head upright) with a measuring rod attached to the balanced Seca scale to the nearest of 0.1 cm. Furthermore, non-stretch tape was used to measure waist circumference (WC), which was measured at the midpoint of the lowest rib and iliac crest at the end of expiration, using a measuring tape to the nearest of 0.1 cm. Hip circumference was measured at the widest point on the buttocks. The waist and hip circumferences were measured to the nearest 0.5 cm. Body mass index (BMI) was calculated by dividing the weight (kg) by height (m) squared. This study adopted the definition of the World Health Organization, in which BMI was categorized as normal weight (18.5ŌĆō24.9 kg/m2), overweight (25ŌĆō29.9 kg/m2) and obese (Ōēź30 kg/m2) [10]. BP (systolic and diastolic) were obtained from the left arm (mm Hg) using a validated mercury sphygmomanometer. BP was measured twice while the patient was seated after relaxing in a quiet environment, with an empty bladder. The average of the two measurements was considered the participantŌĆÖs BP.
PA level was evaluated using the IPAQ-short version [11]. It has been used as an evaluation tool for PA among adults (aged 15ŌĆō69 years) to estimate weekly calorie expenditure expressed as metabolic equivalents per week (METs/wk). The total IPAQ score was calculated by the summation of the duration (in minutes) and frequency (days) of walking and moderate and vigorous activities multiplied by 3.3, 4.0, and 8.0 METs, respectively. According to the IPAQ scoring system, cases were classified as insufficiently active (IA) <600 METs/wk, sufficiently active (SA) 600ŌĆō1,500 METs/wk, and very active (VA) Ōēź1,500 METs/wk.
Perceived stress status was assessed on the day after coronary angiography by using the PSS, which has been translated and validated in Arabic language [12]. It measures the degree to which an individual experienced stress in their life during the previous month. It is a 14-item, 5-point Likert scale (0, never; 1, almost never; 2, sometimes; 3, fairly often; and 4, often). The detail items of the PSS scoring system are items 4, 5, 6, 7, 9, 10, and 13 as reversing scores (0=4, 1=3, 2=2, 3=1, and 4=0). The range of possible PSS scores were from 0 to 56.
The Arabic version of the PSS and PA questionnaires were used, with an intra-correlation coefficient of 0.90 and 0.8, respectively [11,12]. The content validity of the Arabic questionnaire was reviewed by a panel of experts. The required changes were made to clear any ambiguity and ensure comprehension by Palestinian participants after the pilot study.
A reliability test was performed for the overall Arabic version of the questionnaire items, where the values of the CronbachŌĆÖs ╬▒ test for PSS and PA were 0.710 and 0.731, respectively, which indicates high and acceptable internal consistency of the data and its validity for model analysis.
After 12 hours of fasting, venous blood samples were collected from all patients on the day after coronary angiography by well-trained and expert nurses. Venous blood (4.0 mL) was drawn into vacationer tubes. Serum was separated immediately, which was was analyzed for fasting plasma glucose, total cholesterol, high-density lipoprotein cholesterol (HDL-C), and triglycerides (TG). A Mindray BS-200 chemistry analyzer (Mindray DS USA Inc., Mahwah, NJ, USA) was used for blood chemistry analysis. Laboratory tests were conducted in a privately licensed laboratory.
Data were analyzed using the IBM SPSS ver. 24.0 (IBM Corp., Armonk, NY, USA). Descriptive statistics (frequencies and cross-tabulations) were used to describe the main features of the data. Chi-square tests were used to assess the first-hand relationships between the response variables and categorical variables. However, an independent Student t-test was used to assess the relationships between the response variables and continuous variables. Univariate binary logistic regression was performed to assess the magnitude of the relationship between variables by odds ratio (OR) with a 95% confidence interval (95% CI). The obtained OR for the severity of CAD was evaluated using the following predictors: PSS was used as continuous and PA level was categorized as IA (<600 METs/wk), SA (600ŌĆō1,500 METs/wk), and VA (Ōēź1,500 METs/wk). The lowest category of PA was used as the reference. Multivariable binary logistic regression was performed using the backward stepwise elimination method at a level of 0.2. The following confounding factors were identified as significant factors in the univariate analysis and put in and adjusted for in the multivariable model: age, sex, marital status, years of education, history of hypertension, systolic BP, family history of CAD, current smoking, dietary supplements, and BMI. Statistical significance was set at P<0.05.
This study included 423 patients who were newly diagnosed with CAD. The CAD diagnosis was confirmed using coronary angiography results. CAD severity was dichotomized by the Gensini score into severe and non-severe, which accounted for 63.8% (270) and 36.2% (153), respectively. The mean age of participants was 56.7 years with a standard deviation (SD) of 5.9. The proportion of severe CAD among men (86.7%) was higher than that among women. The majority (95.2%) of patients with severe CAD were married. About two-thirds (71.2%) of the participants had only a school education, and 66.3% of them had severe CAD compared with those with university education.
Regarding monthly income, 71.6% of the participants were below the poverty line Ōēż$550; however, the distribution of CAD severity between low and high monthly income was relatively equal. In addition, 56% of all participants had a family history of CAD, and 58.9% of them had severe CAD. More than half (51.5%) of the participants were current smokers, and 61.5% of them had severe CAD. Concerning comorbidities, 57.9% of the participants had a history of hypertension, and 54.1% of them had severe CAD. The overall mean┬▒SD systolic BP of participants was 138.94┬▒18.57 mm Hg, which was higher among severe CAD 141.75┬▒18.38 mm Hg compared with among non-severe CAD cases. Approximately 48.7% of the participants had a history of DM, and 49.3% of them were classified as having severe CAD. Majority of the participants (96.5%) did not use dietary supplements. Moreover, our results revealed that 45.2% and 46.5% of all participants were overweight and obese, respectively, and the percentage of obese individuals was much higher (74.1%) among severe CAD cases than among non-severe cases (25.9%). Additionally, the mean WC was greater in the severe CAD group than in the non-severe group (Table 1).
The relationships between CAD severity and the distributions of categorical independent variables were assessed using the chi-square test. The findings revealed significant statistical associations among sex, marital status, educational level, smoking status, history of hypertension, use of dietary supplements, and BMI categories comparing severe CAD with non-severe. An independent Student t-test was used for continuous variables, which detected significant differences among the mean systolic BP, BMI, and WC. Furthermore, our results showed that the mean differences in lipid profiles and glucose levels among patients with severe CAD were not significantly different from those in patients with non-severe CAD (Table 1).
The results in Table 2 demonstrate that the mean┬▒SD total PSS score of the patients was 27.86┬▒4.57. The score was slightly higher among the severe CAD group (28.22┬▒4.53) compared with among the non-severe (27.86┬▒4.61); this difference was statistically significant between the CAD severity groups (P=0.034). On the other hand, the results in Table 2 show that the percentages of SA, active, and IA categories of PA were higher among severe CAD cases than among non-severe CAD cases. These differences were statistically significant (P<0.001). Moreover, because of known sex variation in CAD [13], subgroup analysis and stratification by sex were conducted. The results illustrated that, in PSS, a statistically significant mean difference was found among women when comparing severe with non-severe CAD cases (P=0.004). However, in the PA categories, a relationship was detected among men when comparing severe with non-severe CAD cases (P=0.001).
We used binary logistic regression to assess the baseline association between PSS, PA levels, and CAD severity, which was dichotomized into severe and non-severe (Table 3). The univariate regression analysis showed that the odds of CAD severity were significantly increased by 5% for each score increase in the PSS (OR, 1.05; 95% CI, 11.02ŌĆō1.09; P=0.035). Regarding the association between PA levels and the severity of CAD, the analysis showed that the odds of severity of CAD among VA patients was decreased by 62% compared with among the IA patients; however, this association was not statistically significant at the 0.05% level (OR, 0.62; 95% CI, 0.37ŌĆō1.07; P=0.088). In contrast, the odds of CAD severity significantly increased by 74% among SA patients compared with among the IA patients (OR, 1.74; 95% CI, 1.02ŌĆō2.97; P=0.040). Moreover, univariate regression analysis revealed that the odds of CAD severity were significantly increased by the following predictors: male sex, being married, university education, current smoking, obesity, increased systolic BP, and WC. However, it decreased with the use of dietary supplements and a history of hypertension of at least 3 months under treatment (Table 3).
Multivariable logistic regression was performed to assess the simultaneous association between the severity of CAD as a response variable and all other independent variables, including PSS and PA, which showed statistical inferences in univariate regression (Table 3). Keeping all other variables constant at baseline, the results showed that PSS, PA level, age, sex, smoking status, history of hypertension, systolic BP, and BMI were predictors of CAD severity. Thus, the odds of CAD severity markedly increased compared with that in univariate regression to 9% when the PSS increased by one score (OR, 1.09; 95% CI, 1.01ŌĆō1.14; P=0.030). Interestingly, the odds of CAD severity among SA individuals were significantly changed in multivariable regression to a protective factor, in which the adjusted OR declined to 43% when comparing SA to IA patients with a significant statistical inference (OR, 0.43; 95% CI, 0.23ŌĆō0.72; P=0.027). In addition, the adjusted OR of CAD severity dropped to 0.54, when comparing VA to SA patients with a notable linear association in protection ability; however, this association was not statistically significant (OR, 0.54; 95% CI, 0.24ŌĆō1.87; P=0.390).
Regarding other confounding variables, the multivariable regression analysis showed that the odds of CAD severity significantly increased by 5% when the age increase by 1 year (OR, 1.05; 95% CI, 1.01ŌĆō1.10; P=0.049). Moreover, the OR of severe CAD among men was 5.64 greater than that among women (OR, 5.64; 95% CI, 2.98ŌĆō10.68; P<0.001). In addition, the odds of CAD severity among current smokers dropped in multivariable regression to 75% compared with that in univariate analysis, in which the association remained significantly higher among smokers compared with among non-smokers (OR, 1.75; 95% CI, 1.01ŌĆō 3.50; P<0.001). Furthermore, the odds of CAD severity significantly increased by 29% when the systolic BP increase by 1 mm Hg (OR, 1.29; 95% CI, 1.14ŌĆō1.44; P<0.001). However, the odds of severe CAD increased by 2.45 times among obese patients compared with among those with normal BMI (OR, 2.45; 95% CI, 1.02ŌĆō5.85; P<0.001). The odds of CAD severity decreased by 48% in patients with a history of hypertension and under treatment for at least 3 months with no history of hypertension (OR, 0.48; 95% CI, 0.25ŌĆō0.79; P=0.005). In addition, a subgroup multivariable logistic regression analysis was conducted to determine the association between CAD severity and sex variation (Table 4). The results showed that the adjusted OR of PSS total score was 27% higher among severe CAD females patients compared with among the non-severe CAD female patients (OR, 1.27; 95% CI, 1.11ŌĆō 1.46; P<0.001); however, no association was found among men. In contrast, the adjusted OR of PA categories was protective by 16% among men when comparing IA to SA (OR, 0.16; 95% CI, 1.06ŌĆō0.34; P=0.035); however, no association was found among women.
This study is among the few studies that explained the association between the severity of CAD and perceived stress status and PA level. The main findings revealed that CAD severity was significantly associated with an increase in perceived stress status. In addition, sufficient PA was associated with CAD severity.
The findings from many meta-analyses studies came to support the association between perceived stress and CVDs in general. However, the association between perceived stress and CAD severity has not yet been sufficiently studied. A meta-analysis suggested that high perceived stress is associated with a moderately increased risk of incident CVDs [14]. In another meta-analysis of prospective observational studies, work-related stress was associated with 40% of CVD events [15]. However, this relationship has been presented as a causal association in some studies, and perceived stress was considered an independent CVD risk factor. The nature of stress requires more analysis based on its theoretical concept and defining the object of measurement to detect whether it is perceived stress that it is measured, or other parameters that relate to it, such as personality factors or merely the affective response to stress.
Our findings are consistent with those of a newly published study conducted in Turkey. The study reported that the odds of CAD severity significantly increased with PSS [16]. The difference from our study was the scale used (SYNTAX) to classify the severity of CAD score, wherein in our study, the Gensini score was used. This was supported by a cross-sectional study conducted by Almuwaqqat et al. [17] among 276 patients with recent myocardial infarction to explore the relationship between mental stress and severity of obstructive CAD using the Gensini score. They found that the odds of CAD severity were significantly increased with mental stress in men but not in women. This was supported by the Stockholm Female Coronary Angiography Study [13], which demonstrated that women with severe stress had accelerated disease progression and decreased luminal diameter of the coronary artery over the 3-year follow-up. Although sex was a significant predictor of CAD severity, as shown in our study, this study only focused on females. Another study from Iran showed that an inverse significant correlation exists between the extent of coronary stenosis and severity of anxiety in patients who underwent coronary angiography [18]. This inverse association was observed because the researchers depended only on the number of coronaries, which could not reflect the extent of CAD severity. Some standardized tools such as the Gensini scores considered one stenosed coronary artery of 70% or more in the proximal left anterior descending artery as severe CAD.
In contrast, our results revealed that CAD severity was inversely associated with sufficient PA. Since there is limited evidence on the association between PA and CAD severity, this study showed that physically active CAD patients tended to be at low risk for mortality [19]. This finding is supported by evidence that regular PA is associated with less severe coronary artery occlusions [20]. The American Heart Association guideline recommendation for patients with CAD is to perform moderate-to-vigorous exercises regularly for at least 30 minutes, 5 days per week, which seems to confer further cardio-protection [21]. In the United States, Weinberg et al. [22] conducted a cross-sectional study among 544 moneposed women to evaluate the risk of PA levels on the severity of atherosclerosis, assessed by the coronary artery calcium score. They showed that increasing PA levels were significantly associated with decreased coronary artery calcium severity. Another study by Mohseni Pouya et al. [23] reported that physically active CAD patients had less arterial stenosis (<50%). Another study showed a significant positive impact of clinical parameters, including PA and regular exercise, on the severity of coronary artery stenosis [24]. Moreover, it is possible to involve changes in lipid metabolism, and evidence has shown that physically active individuals have higher HDL-C by 20%ŌĆō30% and slightly lower TG levels than physically inactive individuals [8].
In a prospective cohort study, Stewart et al. [21] assessed the association of the severity of coronary artery calcium score among 13,977 patients within 10 years of follow-up, and reported that the total coronary artery calcium score was significantly higher among the group reporting a low frequency of PA.
Regarding other independent predictors of CAD severity, our analysis showed that the mean age of patients was 56.7 years, which significantly increased the odds of CAD severity. This result was consistent with a systematic review published in 2015 that revealed that the mean age of hospitalized patients presenting with ACS in three different studies was 55, 58.2, and 61.2 years [25]. Concerning Middle Eastern countries, a recent study reported that the median age of hospitalized patients who present with ACS was 56 years [26]. However, the mean age of CAD in Palestine was much lower, as it reported a mean of 47.4 years among Gaza citizens of both genders [3]. Moreover, our analysis showed that the severity of CAD was significantly associated with obesity. This result is consistent with the results of this study, which reported a positive association between increased BMI Ōēź25 kg/m2 and severity of CAD [27]. In contrast, another study reported a negative correlation between CAD severity and an increased BMI [28]. This can be explained by varying ethnicity, influencing bodybuilding, and differences in BMI between countries.
Furthermore, we found that systolic BP >140 mm Hg was able to predict the severity of CAD. This finding was consistent with a study from Turkey that reported that high brachial systolic BP was a significant predictor for the severity of CAD classified by both SYNTAX and Gensini score [29]. However, a history of hypertension was strongly associated with CAD severity in the present study. This finding is in contrast to a study that reported that a greater risk for CAD severity was positively associated with the presence of hypertension [24]. This may be explained by the variation among study populations in terms of their BP-controlling state during the disease period. It appears that the BP of our participants with hypertension was more controlled by antihypertensive treatment. Additionally, our results showed that smoking is a critical predictor of CAD severity. One study reported that smokers with CAD had a greater risk of CAD severity, as approved by the Gensini and coronary calcification scores [30].
Our study has some limitations. Using interview-based questionnaires to assess patientsŌĆÖ perceived stress and PA could be a potential limitation of the current study. We assessed both PSS 1 month before and PA status 1 week before disease onset. We believe that CAD causes great stress and may limit patientsŌĆÖ PA. High PSS and low PA could be the result of CAD, rather than the cause. For these reasons, the researchers intended to exclude well-known patients with CAD (poststent or post-CABG), renal disease, hepatic dysfunction, anemia or blood disorders, and familial hyperlipidemia, who might have their PSS and PA status affected by the disease. As a result, we selected our cases from all newly discovered CAD patients aged between 35 and 65 years who presented with attacks of chest pain and underwent coronary angiography. Further, we assessed PSS and PA at 1 month and 1 week before the onset of disease, respectively, since those patients were completely healthy with no history of cardiac disease or previous attaches of chest pain during the intended period. However, the main strength of this study is that it is the first to predict the relationship between perceived stress, PA, and the severity of CADŌĆöand not CAD in general.
In conclusion, the odds of CAD severity were positively associated with an increase in the perceived stress status. Sufficient PA was inversely associated with severe CAD. However, no significant associations were found between CAD severity and PA.
ACKNOWLEDGMENTS
We gratefully thank the interventional cardiologists in the Coronary Care Unit at the Al-Shifa Medical Complex for their great contributions to this study.
Figure.┬Ā1.
Flowchart of patients selection. CAD, coronary artery disease; CABG, coronary artery bypass graft.
Table┬Ā1.
Baseline characteristics of the study population by the severity of CAD
| Characteristic | CAD | Severe CAD | Non-severe CAD | P-value* |
|---|---|---|---|---|
| Total | 423 (100.0) | 270 (63.8) | 153 (36.2) | |
| Age (y) | 56.7┬▒5.9 | 57┬▒5.9 | 56┬▒6 | 0.111 |
| Gender | 0.001 | |||
| ŌĆāMale | 314 (74.2) | 234 (86.7) | 80 (52.3) | |
| ŌĆāFemale | 109 (25.8) | 36 (13.3) | 73 (47.7) | |
| Marital status | 0.001 | |||
| ŌĆāMarried | 387 (91.5) | 257 (95.2) | 130 (85) | |
| ŌĆāUnmarried | 36 (8.5) | 13 (4.8) | 23 (15) | |
| Educational level | 0.004 | |||
| ŌĆāSchool education | 301 (71.2) | 179 (66.3) | 112 (79.7) | |
| ŌĆāUniversity education | 122 (28.8) | 91 (33.7) | 31 (20.3) | |
| Monthly income ($) | 0.823 | |||
| ŌĆāŌēż550 | 303 (71.6) | 192 (71.1) | 111 (72.5) | |
| ŌĆā>550 | 120 (28.4) | 78 (28.9) | 42 (27.5) | |
| Family history of CAD | 0.115 | |||
| ŌĆāYes | 237 (56) | 159 (58.9) | 78 (51) | |
| ŌĆāNo | 186 (44) | 111 (41.1) | 75 (49) | |
| History of current smoking | 0.001 | |||
| ŌĆāYes | 218 (51.5) | 166 (61.5) | 52 (34) | |
| ŌĆāNo | 205 (48.5) | 104 (38.5) | 101 (66) | |
| History of hypertension | 0.040 | |||
| ŌĆāYes | 245 (57.9) | 146 (54.1) | 99 (64.7) | |
| ŌĆāNo | 178 (42.1) | 124 (45.9) | 54 (35.3) | |
| Systolic blood pressure (mm Hg) | 138.94┬▒18.57 | 141.75┬▒18.38 | 133.97┬▒17.90 | 0.001 |
| History of diabetes mellitus | 0.763 | |||
| ŌĆāYes | 206 (48.7) | 133 (49.3) | 73 (47.7) | |
| ŌĆāNo | 217 (51.3) | 137 (50.7) | 80 (52.3) | |
| Use aspirin medication | 0.106 | |||
| ŌĆāYes | 215 (50.8) | 129 (47.8) | 86 (56.2) | |
| ŌĆāNo | 208 (49.2) | 141 (52.2) | 67 (43.8) | |
| Use beta-blocker medication | 0.102 | |||
| ŌĆāYes | 177 (41.8) | 121 (44.8) | 56 (36.6) | |
| ŌĆāNo | 246 (58.2) | 149 (55.2) | 97 (63.4) | |
| Use statin medication | 0.913 | |||
| ŌĆāYes | 131 (31) | 83 (30.7) | 48 (31.4) | |
| ŌĆāNo | 292 (69) | 187 (69.3) | 105 (68.6) | |
| Use dietary supplements | 0.025 | |||
| ŌĆāYes | 15 (3.5) | 5 (1.9) | 10 (6.5) | |
| ŌĆāNo | 408 (96.5) | 265 (98.1) | 143 (93.5) | |
| Body mass index (kg/m2) | 0.001 | |||
| ŌĆāNormal | 35 (8.3) | 20 (7.4) | 15 (9.8) | |
| ŌĆāOverweight | 191 (45.2) | 104 (38.5) | 87 (56.9) | |
| ŌĆāObesity | 197 (46.5) | 146 (54.1) | 51 (33.3) | |
| Waist circumference (cm) | 108.54┬▒11.01 | 110.10┬▒11.04 | 105.78┬▒10.34 | 0.001 |
| Fasting blood glucose (mg/dL) | 122.56┬▒44.74 | 124.34┬▒45.28 | 118.59┬▒43.67 | 0.204 |
| Cholesterol (mg/dL) | 179.86┬▒42.44 | 181.12┬▒41.77 | 176.20┬▒39.15 | 0.234 |
| Triglyceride (mg/dL) | 163.39┬▒59.56 | 161.7┬▒56.65 | 166.37┬▒64.48 | 0.439 |
| High-density lipoprotein cholesterol (mg/dL) | 41.40┬▒6.15 | 41.28┬▒5.87 | 41.62┬▒6.65 | 0.587 |
| Low-density lipoprotein cholesterol (mg/dL) | 105.12┬▒38.80 | 107.52┬▒38.99 | 100.52┬▒38.51 | 0.067 |
Table┬Ā2.
Severity of CAD by PSS and physical activity levels by gender
| Variable | CAD | Severe CAD | Non-severe CAD | P-value* |
|---|---|---|---|---|
| Gender | ||||
| ŌĆāPSS | 27.86┬▒4.57 | 28.22┬▒4.53 | 27.24┬▒4.61 | 0.034 |
| ŌĆāPA categories (METs/wk) | 0.001 | |||
| ŌĆāŌĆāIA (<600) | 93 (22.0) | 58 (21.5) | 35 (22.9) | |
| ŌĆāŌĆāSA (600ŌĆō1,500) | 187 (44.2) | 139 (51.5) | 48 (31.4) | |
| ŌĆāŌĆāVA (Ōēź1,500) | 143 (33.8) | 73 (27.0) | 70 (45.8) | |
| Males | ||||
| ŌĆāPSS | 27.72┬▒4.49 | 27.92┬▒4.50 | 27.15┬▒4.43 | 0.182 |
| ŌĆāPA categories (METs/wk) | 0.001 | |||
| ŌĆāŌĆāIA (<600) | 60 (19.1) | 44 (18.8) | 16 (20.0) | |
| ŌĆāŌĆāSA (600ŌĆō1,500) | 149 (47.5) | 125 (53.4) | 24 (30.0) | |
| ŌĆāŌĆāVA (Ōēź1,500) | 105 (33.4) | 65 (27.8) | 40 (50.0) | |
| Females | ||||
| ŌĆāPSS | 28.26┬▒4.80 | 30.13┬▒4.25 | 27┬▒4.82 | 0.004 |
| ŌĆāPA categories (METs/wk) | 0.133 | |||
| ŌĆāŌĆāIA (<600) | 33 (30.2) | 14 (38.9) | 19 (26.0) | |
| ŌĆāŌĆāSA (600ŌĆō1,500) | 38 (34.9) | 14 (38.9) | 24 (32.9) | |
| ŌĆāŌĆāVA (Ōēź1,500) | 38 (34.9) | 8 (22.2) | 30 (41.1) |
Table┬Ā3.
Binary logistic regression of severity of CAD (severe/non-severe) dependent to Perceived Stress Scale and PA levels
| Variable |
Univariate analysis |
Multivariable analysis |
||
|---|---|---|---|---|
| Unadjusted OR (95% CI) | P-value | Adjusted OR (95% CI) | P-value* | |
| PSS total score | 1.05 (1.02ŌĆō1.09) | 0.035 | 1.09 (1.01ŌĆō1.14) | 0.030 |
| PA categories (METs/wk) | ||||
| ŌĆāInsufficiently active (<600) | (Reference) | - | - | - |
| ŌĆāSufficiently active (600ŌĆō1,500) | 1.74 (1.02ŌĆō2.97) | 0.040 | 0.43 (0.23ŌĆō0.72) | 0.027 |
| ŌĆāVery active (Ōēź1,500) | 0.62 (0.37ŌĆō1.07) | 0.088 | 0.54 (0.24ŌĆō1.87) | 0.390 |
| Age (y) | 1.02 (0.99ŌĆō1.06) | 0.117 | 1.05 (1.01ŌĆō1.10) | 0.049 |
| Gender (male) | 5.39 (3.69ŌĆō9.51) | 0.001 | 5.64 (2.98ŌĆō10.68) | 0.001 |
| Marital status (married) | 3.49 (1.17ŌĆō7.12) | 0.001 | - | - |
| Education level (y) | 2.00 (1.25ŌĆō3.19) | 0.004 | - | - |
| Family history of CAD | 1.37 (0.92ŌĆō2.05) | 0.116 | - | - |
| Smoking status smokers | 3.10 (2.04ŌĆō4.69) | 0.001 | 1.75 (1.01ŌĆō3.50) | 0.001 |
| History of hypertension | 0.64 (0.42ŌĆō0.96) | 0.034 | 0.48 (0.25ŌĆō0.79) | 0.005 |
| Systolic blood pressure (mm Hg) | 1.24 (1.12ŌĆō1.36) | 0.001 | 1.29 (1.14ŌĆō1.44) | 0.001 |
| Use dietary supplements | 0.27 (0.09ŌĆō0.80) | 0.019 | - | - |
| Waist circumference (cm) | 1.03 (1.01ŌĆō1.05) | 0.001 | - | - |
| Body mass index | ||||
| ŌĆāNormal | (Reference) | - | - | - |
| ŌĆāOverweight | 0.89 (0.43ŌĆō1.85) | 0.769 | 0.96 (0.41ŌĆō2.84) | 0.786 |
| ŌĆāObesity | 2.14 (1.02ŌĆō4.50) | 0.043 | 2.45 (1.02ŌĆō5.85) | 0.001 |
Table┬Ā4.
Subgroup analysis: multivariable logistic regression of severity of CAD by gender
REFERENCES
1. Pimple P, Lima BB, Hammadah M, Wilmot K, Ramadan R, Levantsevych O, et al. Psychological distress and subsequent cardiovascular events in individuals with coronary artery disease. J Am Heart Assoc 2019;8:e011866.



2. World Health Organization. Cardiovascular diseases (CVDs): key facts. Geneva: World Health Organization; 2017.
3. Jamee Shahwan A, Abed Y, Desormais I, Magne J, Preux PM, Aboyans V, et al. Epidemiology of coronary artery disease and stroke and associated risk factors in Gaza community: Palestine. PLoS One 2019;14:e0211131.



4. Marie M, Hannigan B, Jones A. Mental health needs and services in the West Bank, Palestine. Int J Ment Health Syst 2016;10:23.



5. Allabadi H. Mental disorders among cardiac disease patients in a Palestinian population. Basel: Universitat Basel; 2019.
6. Katsarou AL, Triposkiadis F, Panagiotakos D. Perceived stress and vascular disease: where are we now? Angiology 2013;64:529-34.



7. Fioranelli M, Bottaccioli AG, Bottaccioli F, Bianchi M, Rovesti M, Roccia MG. Stress and inflammation in coronary artery disease: a review psychoneuroendocrineimmunology-based. Front Immunol 2018;9:2031.



8. Wang Y, Xu D. Effects of aerobic exercise on lipids and lipoproteins. Lipids Health Dis 2017;16:132.




9. Neeland IJ, Patel RS, Eshtehardi P, Dhawan S, McDaniel MC, Rab ST, et al. Coronary angiographic scoring systems: an evaluation of their equivalence and validity. Am Heart J 2012;164:547-52.



10. Kulkarni P, Ashok NC, Kumar DS, Siddalingappa H, Madhu B. World health organization: body mass index for age criteria as a tool for prediction of childhood and adolescent morbidity: a novel approach in southern Karnataka, India. Int J Prev Med 2014;5:695-702.


11. Craig CL, Marshall AL, Sjostrom M, Bauman AE, Booth ML, Ainsworth BE, et al. International physical activity questionnaire: 12-country reliability and validity. Med Sci Sports Exerc 2003;35:1381-95.


12. Almadi T, Cathers I, Hamdan Mansour AM, Chow CM. An Arabic version of the perceived stress scale: translation and validation study. Int J Nurs Stud 2012;49:84-9.


13. Wang HX, Leineweber C, Kirkeeide R, Svane B, Schenck-Gustafsson K, Theorell T, et al. Psychosocial stress and atherosclerosis: family and work stress accelerate progression of coronary disease in women: the Stockholm Female Coronary Angiography Study. J Intern Med 2007;261:245-54.


14. Richardson S, Shaffer JA, Falzon L, Krupka D, Davidson KW, Edmondson D. Meta-analysis of perceived stress and its association with incident coronary heart disease. Am J Cardiol 2012;110:1711-6.



15. Steptoe A, Kivimaki M. Stress and cardiovascular disease: an update on current knowledge. Annu Rev Public Health 2013;34:337-54.


16. Kahraman S, Cam Kahraman F, Zencirkiran Agus H, Kalkan AK, Uzun F, Karakayali M, et al. Perceived stress level is associated with coronary artery disease severity in patients with ST-segment elevation myocardial infarction. Turk Kardiyol Dern Ars 2020;48:494-503.


17. Almuwaqqat Z, Sullivan S, Hammadah M, Lima BB, Shah AJ, Abdelhadi N, et al. Sex-specific association between coronary artery disease severity and myocardial ischemia induced by mental stress. Psychosom Med 2019;81:57-66.



18. Assari S, Zandi H, Ahmadi K, Kazemi Saleh D. Extent of coronary stenosis and anxiety symptoms among patients undergoing coronary angiography. J Tehran Heart Cent 2017;12:155-9.


19. Tian X, Du H, Li L, Bennett D, Gao R, Li S, et al. Fruit consumption and physical activity in relation to all-cause and cardiovascular mortality among 70,000 Chinese adults with pre-existing vascular disease. PLoS One 2017;12:e0173054.



20. Najafi M, Sheikhvatan M. Gender differences in coronary artery disease: correlational study on dietary pattern and known cardiovascular risk factors. Int Cardiovasc Res J 2013;7:124-9.


21. Stewart RA, Held C, Armstrong PW, Cannon CP, Hadziosmanovic N, Hagstrom E, et al. Associations Between the amount and intensity of physical activity and major cardiovascular events in patients with stable coronary heart disease: results from the STABILITY Trial. Circulation 2015;132(suppl_3):A13864.
22. Weinberg N, Young A, Hunter CJ, Agrawal N, Mao S, Budoff MJ. Physical activity, hormone replacement therapy, and the presence of coronary calcium in midlife women. Women Health 2012;52:423-36.



23. Mohseni Pouya H, Hajimiri K, Esmaeili Shahmirzadi S, Golshani S, Amrei H, Seifi Makrani A. Relationship between health promoting behaviors and severity of coronary artery stenosis in angiography department in Mazandaran Heart Center. J Mazandaran Univ Med Sci 2015;25:19-29.
24. Pouya HM, Shahmirzadi SE, Hajimiri K, Majlessi F, Bagheri M. The relationship between clinical parameters and lifestyle variables among patients with severity of coronary artery stenosis: a cross-sectional analysis based on the severity of coronary artery stenosis. J Educ Health Promot 2017;6:66.



25. Pasupathy S, Air T, Dreyer RP, Tavella R, Beltrame JF. Systematic review of patients presenting with suspected myocardial infarction and nonobstructive coronary arteries. Circulation 2015;131:861-70.


26. Traina MI, Almahmeed W, Edris A, Murat Tuzcu E. Coronary heart disease in the Middle East and North Africa: current status and future goals. Curr Atheroscler Rep 2017;19:24.



27. Gerami H, Javadi M, Hosseini SK, Maljaei MB, Fakhrzadeh H. Coronary artery stenosis and associations with indicators of anthropometric and diet in patients undergoing coronary angiography. J Diabetes Metab Disord 2018;17:203-10.




28. Dores H, de Araujo Goncalves P, Carvalho MS, Sousa PJ, Ferreira A, Cardim N, et al. Body mass index as a predictor of the presence but not the severity of coronary artery disease evaluated by cardiac computed tomography. Eur J Prev Cardiol 2014;21:1387-93.



- TOOLS







